Clinical Rounds: Hyperaldosteronism in a 10-year-old domestic shorthaired cat
A 10-year-old spayed female domestic shorthaired cat was presented with polyuria, kidney disease and an abdominal tumor. See how the Clinical Rounds team produced a life-saving outcome in this case.
The Clinical Rounds team is from the Department of Small Animal Clinical Sciences, College of Veterinary Medicine, The University of Tennessee, Knoxville, Tennessee.
Clinical rounds coordinator: Jeanne Lane, DVM, DACVIM (oncology)
Internal medicine: Amy Holford, VMD, DACVIM
Radiology: Mylene Auger, DVM
Soft tissue surgery: Whitney Degroot, DVM
Anatomic pathology: Lani Bower, DVM
Medical oncology: Lark Walters, DVM
Primary hyperaldosteronism (also known as Conn's syndrome) is the most common adrenocortical disorder seen in cats,1 and it is suspected to be underrecognized. Hyperaldosteronism is seen in middle-aged to older cats,2 and clinical signs are due to high aldosterone concentrations secreted by an adrenocortical tumor, resulting in hypokalemia and systemic hypertension. Most cats have a unilateral adrenal mass, and both carcinomas and adenomas have been reported.1
CASE PRESENTATION

Figure 1: The 10-year-old spayed female domestic shorthaired cat featured in this case. All images courtesy of the University of Tennessee. A 10-year-old spayed female domestic shorthaired cat (Figure 1) was presented to the University of Tennessee (UT) Community Practice service for a wellness examination. The cat had been recently adopted, and polyuria was noted. On physical examination the cat was overweight (body condition score 8/9); the exam was otherwise unremarkable. A complete blood count, serum chemistry profile with electrolytes, and urinalysis were performed. The only significant abnormality was a urine specific gravity of 1.022, indicating potential renal insufficiency. Systolic blood pressure measured via Doppler was 160 mm Hg. A weight loss plan and increased exercise were recommended.
Three months later, the cat was presented to the Community Practice for further investigation of progressive polyuria and polydipsia. The owners reported that the water bowl required refilling three to four times per day. On physical examination the cat had lost 0.7 lb on the diet previously recommended. A grade III/VI left parasternal heart murmur was auscultated. The cat's systolic blood pressure was 165 mm Hg. A complete blood count revealed a mild normocytic, normochromic anemia of 30.1% (reference range = 34% to 46%). A serum chemistry profile with electrolytes indicated hypernatremia of 158 mmol/L (reference range = 141 to 147 mmol/L); potassium was near the low end of normal at 2.9 mmol/L (reference range = 2.5 to 4.6 mmol/L). Urinalysis revealed a urine specific gravity of 1.013. Urine culture results were negative. The total thyroxine (T4) concentration was normal at 2.2 µg/dl (reference range = 1 to 4 µg/dl). A symmetric dimethylarginine (SDMA) concentration was elevated at 17 µg/dl (reference range = 0 to 14 µg/dl).
Given confirmation of chronic kidney disease, a therapeutic renal diet was recommended as well as a recheck of blood pressure to determine if anti-hypertensive medication was indicated. Abdominal imaging was also recommended, which was pursued several weeks later. An abdominal ultrasonographic examination identified a 2-x-2.5-cm, round, well-defined hypoechoic mass in the region of the left adrenal gland. The mass was in contact with the vena cava at the entrance of the phrenicoabdominal vein. Additionally, evidence of bilateral chronic kidney disease with many calculi and evidence of infarcts was noted.
Tumor staging and treatment
Given the finding of a left adrenal mass in conjunction with borderline hypokalemia and systemic hypertension, hyperaldosteronism was suspected. A serum aldosterone concentration was found to be elevated at > 1,650 pg/ml (reference range = 3.3 to 261.3 pg/ml). Potassium gluconate was prescribed (2 mEq orally twice daily). Surgical excision of the left adrenal gland was recommended.
One week later, preoperative electrolytes showed improvement in potassium concentration (3.6 mmol/L). Thoracic radiographs revealed no evidence of pulmonary metastatic disease. The cat was transferred to the UT Soft Tissue Surgery service for left adrenalectomy. In surgery, the mass was noted to have invaded the phrenicoabdominal vein to a mild extent. There were no surgical complications and recovery was smooth. Postoperative electrolyte concentrations were stable. A serum aldosterone concentration was normal at 110 pg/ml. Histopathologic evaluation of the adrenal mass confirmed a cortical carcinoma, consistent with hyperaldosteronism.
Follow-up
One month later, the cat was presented to the Community Practice service for a postoperative recheck. The polyuria and polydipsia had reportedly resolved. The cat's blood pressure was normal at 125 mm Hg. The results of a complete blood count were normal. A serum chemistry profile with electrolytes showed continued mild hypernatremia (153 mmol/L) and mild hyperkalemia (4.9 mmol/L). A serum aldosterone concentration was normal at 69.3 pg/ml. Urinalysis showed a urine specific gravity of 1.035 and the SDMA concentration was 19 µg/dl.
Oral potassium supplementation was discontinued, and continued management of chronic kidney disease was recommended. Re-evaluation every three months was recommended for continued monitoring of kidney function.
References
1. Daniel G, Mahony OM, Marchovich JE, et al. Clinical findings, diagnostics, and outcome in 33 cats with adrenal neoplasia (2002-2013). J Fel Med Surg 2014;18:77-84.
2. Lo AJ, Holt DE, Brown DC, et al. Treatment of aldosterone-secreting adrenocortical tumors in cats by unilateral adrenalectomy: 10 cases (2002-2012). J Vet Intern Med 2014;28:137-143.
INTERNAL MEDICINE PERSPECTIVE
Amy Holford, VMD, DACVIM
Primary hyperaldosteronism is also known as Conn's disease or Conn's syndrome. There are three components to this disorder that result when excess aldosterone is produced:
1. Systemic hypertension
2. Increased urinary loss of potassium, resulting in profound hypokalemia
3. Metabolic alkalosis due to intracellular hydrogen shifts and urinary losses
The primary role of aldosterone, which is produced in the zona glomerulosa of the adrenal cortex, is to regulate blood pressure and maintain homeostasis of the extracellular fluid volume in response to changes in hemodynamics and electrolytes. Aldosterone secretion is regulated by the renin-angiotensin-aldosterone system (RAAS) and extracellular potassium concentrations. Aldosterone increases secretion of potassium and hydrogen and resorption of sodium and chloride in the distal nephrons. Increases in aldosterone will increase sodium and extracellular fluid volume, therefore restoring blood pressure and blood volume.
The most common clinical signs described in cats with primary hypoaldosteronism include weakness, cervical ventroflexion and lethargy. Most affected cats are geriatric. Weakness can be acute or chronic in nature. Hind limb weakness, forelimb stiffness and dysphagia have also been described.1 Signs associated with systemic hypertension such as retinal hemorrhage or detachment, cardiac changes (heart murmur) and renal changes (polyuria/polydipsia) may also be seen.
Biochemical findings typically include moderate to severe hypokalemia, which tends to develop later in the course of disease, a normal to mildly increased sodium concentration, and increased creatine kinase activity secondary to hypokalemic myopathy.
The hallmarks of primary hyperaldosteronism include hypertension, hypokalemia, hyperaldosteronemia, and the presence of an adrenal mass. Primary hyperaldosteronism should be suspected in cats with hypertension and hypokalemia when another cause cannot be identified.1
Serum aldosterone concentrations can be performed at many veterinary laboratories and the results must be interpreted in light of potassium concentrations.1 Because hypokalemia is a potent suppressant of aldosterone secretion in normal animals, cats with low potassium and “normal” aldosterone concentrations may actually be considered abnormal given the circumstances.
Secondary causes of increased aldosterone can be related to renal disease, cardiovascular disease or liver failure.1 These causes are a result of increased renin, and aldosterone values are typically lower than those seen with primary disease. Renin concentrations are not currently easily assessed in the United States, so most clinicians rely on aldosterone measurement as a solitary test.1
A thorough history, physical examination and minimum database will rule out most other causes of hypokalemia in cats. These can include renal failure, hepatic dysfunction, infection, gastrointestinal disease, cardiac disease and endocrinopathies such as hyperthyroidism and diabetes mellitus. Similarly, other causes of systemic hypertension such as renal failure and hyperthyroidism must be ruled out. It is important to keep in mind that cats may develop hypokalemia later in the course of the disease, so if hypertension is documented and an obvious underlying cause such as cardiovascular disease or hyperthyroidism is not identified, primary hyperaldosteronism should be considered. Abdominal imaging will confirm the presence of an adrenal mass in cats with primary hyperaldosteronism.
Although surgical management is the treatment of choice in cats with primary hyperaldosteronism, treatment in the preoperative period will certainly make them better anesthetic and surgical candidates. If a patient is unable to undergo surgical intervention for whatever reason, medical management is a viable option. Medically treated cats have been reported to survive from many months to years.1 Components of medical management include potassium supplementation, anti-hypertensive agents and aldosterone antagonist therapy.
> Potassium supplementation: Potassium gluconate, most commonly in the form of Tumil K (potassium gluconate) or RenaKare (potassium gluconate), should be administered at 2 mEq/4.5 kg orally in food twice daily.2 Adjustments should be made according to monitored potassium concentrations. Tumil K (Virbac) and RenaKare (Neogan) are available in flavored powders (2 mEq per ¼ tsp), tablets (2 mEq/tablet) and gels (2 mEq/¼ tsp). Weekly monitoring is recommended until potassium concentrations are around 4 mmol/L.
> Anti-hypertensive agents: The most common first-line anti-hypertensive agent used in cats is amlodipine. Amlodipine is a calcium channel blocker, inhibiting calcium influx across cell membranes in both cardiac and vascular smooth muscle. It has a greater effect on vascular smooth muscle, thereby acting as a peripheral arteriolar vasodilator and reducing afterload. Amlodipine also depresses impulse formation and conduction velocity in cardiac muscle. The recommended starting dose is 0.625 mg/cat orally once daily.2 This can be accomplished by splitting a 2.5-mg tablet into quarters or having the drug compounded into smaller capsules or an oral suspension. The dosage should be adjusted according to blood pressure readings with the goal of achieving a systolic blood pressure of < 160 mm Hg. Increase in increments of 0.625 mg up to 2.5 mg/cat/day. Adverse effects are uncommon. Rarely, cats can develop gingival hyperplasia.
> Aldosterone antagonist therapy: The current drug of choice to inhibit aldosterone is spironolactone. Spironolactone competitively inhibits aldosterone in the distal renal tubules, which results in increased excretion of sodium, chloride and water as well as decreased excretion of potassium, ammonium, phosphate and titratable acid. Spironolactone has no effect on carbonic anhydrase or renal transport mechanisms and has its greatest effect in patients with hyperaldosteronism. The recommended starting dose is 1 to 2 mg/kg orally every 12 hours.2 Most cats require starting with 6.25 mg (1/4 of a 25-mg tablet). Adverse effects are uncommon. Monitor for facial dermatitis and gastrointestinal or central nervous system effects.
Take-home points:
1. Blood pressure monitoring should be routinely performed on all cats. It is best to start when they are young to ensure they are accustomed to having their blood pressure measured and to provide a baseline for comparison in the future.
2. Continued monitoring of trends in blood pressure and potassium and sodium concentrations may lead to earlier diagnosis of primary hyperaldosteronism.
3. A finding of severe hypokalemia should immediately prompt blood pressure assessment, and cats with severe hypertension should have their potassium concentration checked.
4. Treating primary hyperaldosteronism (surgically or medically) will help decrease ongoing damage to the kidneys.
5. Surgery is the treatment of choice for cats with primary hyperaldosteronism. However, medical management has been reported to result in similar survival times in some cats.
References
1. Schulman RL. Feline primary hyperaldosteronism. Vet Clin North Am Small Anim Pract 2010;40:353-359.
2. Plumb DC. Veterinary Drug Handbook. 8th ed. Ames, Iowa: Wiley-Blackwell, 2015.
RADIOLOGY PERSPECTIVE
Mylene Auger, DVM
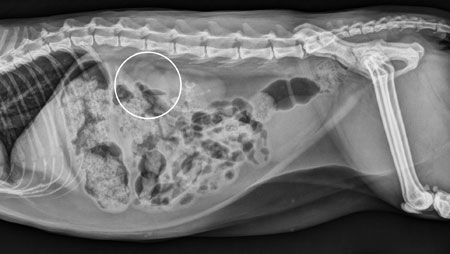
Figure 1: A right lateral radiograph from the patient in this case. There is an ovoid, relatively well-defined soft tissue opaque mass in the retroperitoneal space, superimposed with the cranial poles of both kidneys, consistent with an adrenal mass (circled). The adrenal gland can be seen in this case because it is enlarged.Several imaging modalities can be useful in the staging and presurgical management of adrenal neoplasia. Because the adrenal glands are usually not seen radiographically due to their small size and superimposition with adjacent structures, abdominal radiographs are of limited utility in the diagnosis of adrenal neoplasia. The adrenal glands can occasionally be visible on radiographs when they are calcified or enlarged (Figure 1).1 Calcification of the adrenal glands has been reported as an incidental finding in up to 30% of normal cats, whereas in dogs this finding has been associated with adrenocortical tumors, especially when associated with adrenal enlargement.2,3 Despite their limitations, radiographs are useful for staging to evaluate for the presence of pulmonary metastatic disease and to assess for secondary effects of adrenal dysfunction such as cardiomegaly in the case of hyperaldosteronism and hepatomegaly or dystrophic mineralization of the soft tissues in the case of hyperadrenocorticism (Figures 2A and 2B).
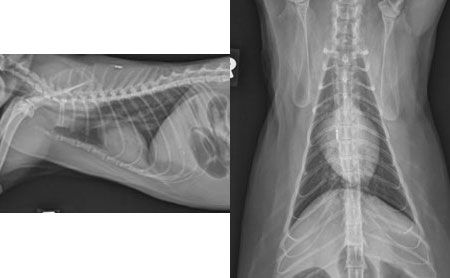
Figures 2A and 2B: Right lateral (left) and ventrodorsal (right) radiographs of the patient in this case showing mild cardiomegaly, which may represent an individual variant or be secondary to cardiac disease (such as hypertrophic cardiomyopathy). Hyperaldosteronism can result in signs of cardiovascular disease due to systemic hypertension. There is no evidence of pulmonary metastatic disease.Ultrasonography is the most commonly used imaging modality in the workup of adrenal disease. This modality is rapid and effective in obtaining information on abdominal organs, is noninvasive and can be performed on animals with little or no sedation. The adrenal glands are located craniomedially to each kidney and are relatively easily visualized using ultrasound, depending on the experience of the sonographer and the quality of the equipment. The most common appearance of the normal feline adrenal gland is well-defined, bean-shaped and hypoechoic with a thin hyperechoic halo (Figure 3).2,3 Fusiform or oval-shaped adrenal glands have also been described.2 The normal height of the adrenal gland can vary between 3 mm and 4.8 mm, and little difference in size and shape is seen between the left and right adrenal glands in cats.2,3
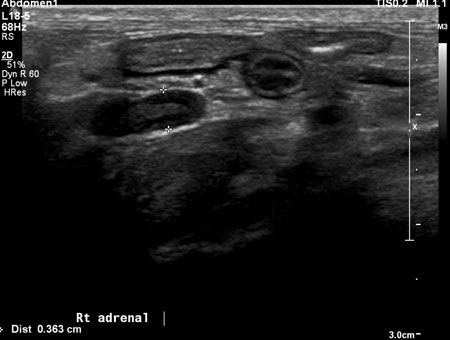
Figure 3: A longitudinal sonographic image from a different cat than the patient in this case illustrating the typical appearance of a normal feline adrenal gland (between cursors). The feline adrenal gland is usually well-defined, bean-shaped and hypoechoic with a thin hyperechoic halo.Primary hyperaldosteronism in cats commonly presents as a unilateral adrenal mass, and three patterns of echogenicity have been reported: hyperechoic and heterogeneous, hypoechoic and homogeneous, and target-like (hyperechoic center with a hypoechoic rim).2 Ultrasonography is useful to determine if the disease is unilateral or bilateral as well as to determine if there is vascular invasion. There are, however, certain limitations associated with sonographic evaluation of adrenal tumors. The contralateral adrenal gland may appear normal sonographically but may be histologically abnormal.2 Invasion into the caudal vena cava is usually easily identified ultrasonographically, with a reported sensitivity and specificity of up to 100% and 96%, respectively, in dogs.4 However, invasion into smaller vessels or into the vessel walls can be difficult to identify, with a sensitivity and specificity of 76% and 96%, respectively, when all vascular invasion is considered (Figures 4A and 4B).4
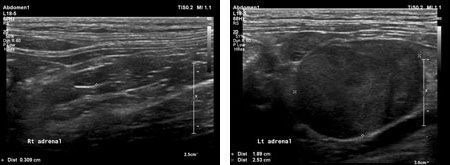
Figures 4A and 4B: Sonographic images from the patient in this case. 4A (left): A longitudinal image of the normal right adrenal gland (between cursors). 4B (right): A longitudinal image of the left adrenal mass (between cursors), which is hypoechoic and measures approximately 2 cm in height by 2.5 cm in length. The mass abuts the caudal vena cava at the entrance of the phrenicoabdominal vein. Blood flow could be seen within the caudal vena cava and phrenicoabdominal vein using color Doppler. The mass was thought to be adhered to the wall of the caudal vena cava but there was no sonographic evidence of vascular invasion.In people, computed tomography (CT) and magnetic resonance imaging (MRI) are the primary modalities used for imaging of adrenal neoplasia as they offer better tumor characterization. They are not influenced by body condition and gastrointestinal gas, and they more readily identify vascular invasion.4,5 In dogs, CT has been shown to be more accurate than ultrasonography in the identification of vascular invasion by adrenal tumors, with reports of up to 92% sensitivity and 100% specificity.5 CT is therefore recommended in the presurgical planning of adrenal neoplasia in dogs.4,5 In cats, only a few case reports describe the use of CT or MRI following ultrasound before adrenalectomy so conclusions as to sensitivity and specificity of these modalities for identification of vascular invasion in cats have not yet been determined (Figure 5).6-8
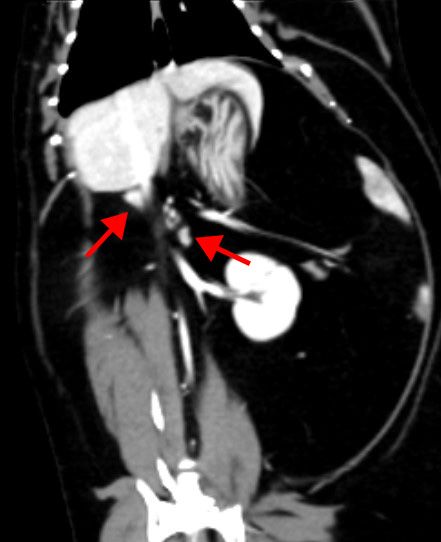
Figure 5: Computed tomography (CT) image from a different cat than the patient in this case showing a post-contrast slightly oblique dorsal plane multiplanar reconstruction of the normal feline adrenal glands (red arrows). The adrenal glands can be found cranially and medially to each kidney.MRI has infrequently been used to further characterize adrenal neoplasia in cats.8 Disadvantages of MRI include the need for general anesthesia as well as longer scan times and increased cost.9 The major advantage of MRI over CT is the superior soft tissue contrast offered by MRI.9 The recommended protocol for adrenal imaging in dogs includes at least two different planes in T2-weighted, T1-weighted and contrast-enhanced T1-weighted sequences.9 These recommendations can be extrapolated to adrenal imaging in cats. However, due to the previously described disadvantages, they are unlikely to become routinely used for this purpose (Figure 6).
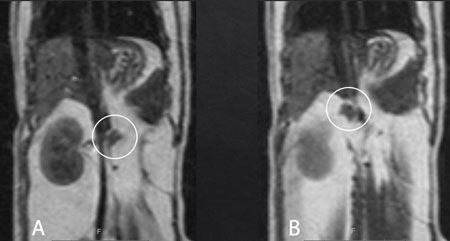
Figure 6: Magnetic resonance imaging (MRI) T1-weighted spin-echo sequence of a different cat than the patient in this case. Dorsal plane images of the normal left (A) and right (B) feline adrenal glands (circled). The adrenal glands can be found cranially and medially to each kidney.
References
1. Tidwell AS, Penninck DG, Besso JG. Imaging of adrenal gland disorders. Vet Clin North Am Small Anim Pract 1997;27:237-254.
2. Combes A, Pey P, Paepe D, et al. Ultrasonographic appearance of adrenal glands in healthy and sick cats. J Feline Med Surg 2013;15:445-457.
3. Mattoon JS, Nyland TG. Adrenal glands. In: Small Animal Diagnostic Ultrasound. 3rd ed. St. Louis, Missouri: Elsevier, 2015.
4. Davis MK, Schochet RA, Wrigley R. Ultrasonographic identification of vascular invasion by adrenal tumors in dogs. Vet Radiol Ultrasound 2012;53:442-445.
5. Schultz RM, Wisner ER, Johnson EG, et al. Contrast-enhanced computed tomography as a preoperative indicator of vascular invasion from adrenal masses in dogs. Vet Radiol Ultrasound 2009;50:625-629.
6. Daniel G, Mahony OM, Markovich JE, et al. Clinical findings, diagnostics and outcome in 33 cats with adrenal neoplasia (2002-2013). J Feline Med Surg 2014;18:77-84.
7. Lo AJ, Holt DE, Brown DC, et al. Treatment of aldosterone-secreting adrenocortical tumors in cats by unilateral adrenalectomy: 10 cases (2002-2012). J Vet Intern Med 2014;28:137-143.
8. Ash RA, Harvey AM, Tasker S. Primary hyperaldosteronism in the cat: a series of 13 cases. J Feline Med Surg 2005;7:173-182.
SURGERY PERSPECTIVE
Whitney DeGroot, DVM
For cats with hyperaldosteronism, a unilateral adrenal mass and no evidence of metastatic disease, surgical excision is the treatment of choice. Successful surgical excision can be curative for both adenomas and adenocarcinomas and also generally resolves systemic hypertension and hypokalemia without the need for continued medical management.1-3
Preoperative therapy should be aimed at controlling hypertension and hypokalemia to improve anesthetic stability. An abdominal computed tomography scan (CT scan) with contrast enhancement can be useful to aid in surgical planning, but its use in feline adrenal imaging has not been well-documented. Therefore, the recommendation for a preoperative CT scan is generally based on surgeon preference; general indications for its use include large or right-sided masses and masses that are vascular or suspected to have vascular invasion on ultrasound.
The adrenal glands are paired glands located in the retroperitoneal space. The left adrenal gland lays craniomedial to the left kidney and is adjacent to the left side of the abdominal aorta. The right adrenal gland is much closer to the right kidney and caudal vena cava, and is typically covered by the caudal extent of the right lateral liver lobe. The phrenicoabdominal veins course ventrally over each gland. The adrenal glands receive their arterial supply from branches arising from the renal, phrenicoabdominal and cranial abdominal arteries. Venous blood collects in sinusoids and drains into the adrenal vein, which empties directly into the vena cava on the right and into the renal vein on the left.4
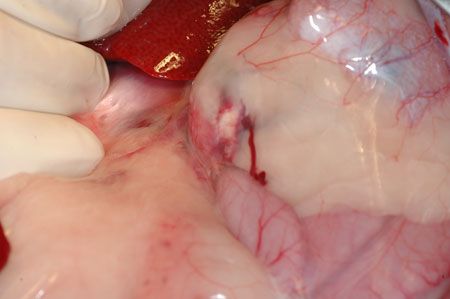
Figure 1: An intraoperative photo from the patient in this case; the left-sided adrenal mass is poorly visualized prior to surgical dissection.Unilateral adrenalectomy can be performed via a ventral midline approach (with or without paracostal extension), a flank approach or laparoscopically.1,4 With a ventral midline approach to left-sided masses, the descending colon is retracted to access the retroperitoneal space. For right-sided masses, the proximal duodenum is retracted and the liver is retracted cranially.3 The adrenal gland can be visualized within the retroperitoneal fat (Figure 1). Blunt dissection using hemostats or forceps and cotton-tipped applicators (Figure 2) is used to dissect the mass without rupturing vascular supply. Blood vessels are ligated using a vessel-sealing device such as the LigaSure (Covidien) (Figure 3) or Hemoclips (Teleflex Medical). Caval thrombectomy may be necessary in cases with vascular invasion and has been successfully reported in cats.5 Major complications of adrenalectomy include surgically induced hemorrhage, postoperative anorexia, lethargy, hepatic lipidosis, anemia, hypotension and sepsis.1,2,6
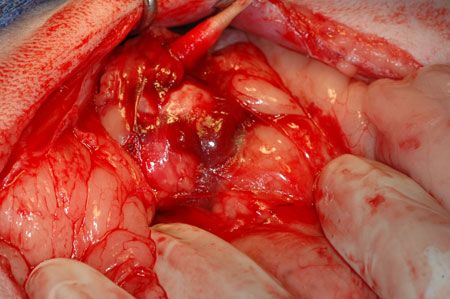
Figure 2: An intraoperative photo from the patient in this case; the use of cotton-tipped applicators can be helpful to dissect delicate surrounding tissues without tearing vascular supply.Recent retrospective analyses of cats undergoing unilateral adrenalectomy for treatment of hyperaldosteronism report survival rates of 60% to 80%.1,2 In both of these case series, all patients that survived to hospital discharge were successfully weaned off anti-hypertensive medications and potassium supplementation within six to eight days after surgery. In one report,1 the median survival time was 1,297 days (range: two to 1,582 days), indicating that patients that survive surgery have good long-term survival. Similarly, in a study in cats with adrenal neoplasia (of any etiology), hypokalemia (potassium < 3 mEq/L) was associated with significantly longer survival times, indicating that cats with hyperaldosteronism due to adrenal neoplasia may have a better prognosis, as compared to those with other tumor types (pheochromocytoma or other cortical tumors).6
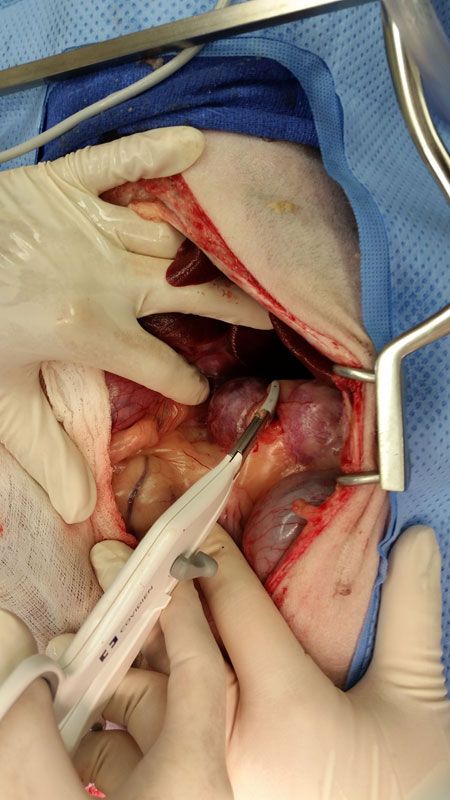
Figure 3: An intraoperative photo from the patient in this case; use of a LigaSure (Covidien) vessel-sealing device to ligate and divide vascular supply to the mass.Postoperative management should include monitoring of systemic blood pressure and serum potassium concentrations. Patients exhibiting hypotension should be examined for evidence of postoperative hemorrhage. Serum potassium concentrations should be monitored, as hyperkalemia can result from hypoaldosteronism caused by excessive negative feedback inhibition on normal aldosterone production. This typically self-corrects over several days after surgery, but moderate to severe hyperkalemia (> 7 mEq/L) should be treated with furosemide. Electrocardiogram should be monitored for the development of arrhythmias secondary to potassium derangement.3
References
1. Lo AJ, Holt DE, Brown DC, et al. Treatment of aldosterone-secreting adrenocortical tumors in cats by unilateral adrenalectomy: 10 cases (2002-2012). J Vet Intern Med 2014;28:137-143.
2. Ash RA, Harvet AM, Tasker S. Primary hyperaldosteronism in the cat: a series of 13 cases. J Feline Med Surg 2005;7:173-182.
3. Monnet E. Adrenal tumors. In Small Animal Soft Tissue Surgery. 1st ed. Ames, Iowa: Wiley-Blackwell, 2013.
4. Tobias KM, Johnson S. Adrenal glands. In Veterinary Surgery: Small Animal. 1st ed. St. Louis, Missouri: Saunders, 2011.
5. Rose SA, Kyles AE, Labelle P, et al. Adrenalectomy and caval thrombectomy in a cat with primary hyperaldosteronism. J Am Anim Hosp Assoc 2007;43:209-214.
6. Daniel G, Mahony OM, Markovicg JE, et al. Clinical findings, diagnostics and outcome in 33 cats with adrenal neoplasia (2002-2013). J Feline Med Surg 2014;18:77-84.
ANATOMIC PATHOLOGY PERSPECTIVE
Lani Bower, DVM
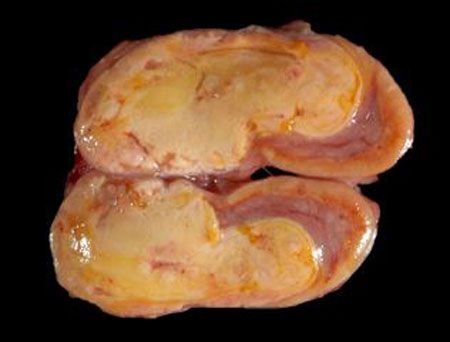
Figure 1: Gross image of an adrenocortical adenoma (not the patient described in this case).Feline primary hyperaldosteronism is most commonly caused by a unilateral functional adrenal cortical adenoma or carcinoma arising from the zona glomerulosa. Neoplasia of the feline adrenal cortex is rare, comprising approximately 0.2% of all reported neoplasms.1 Carcinomas are reported at a slightly higher rate than adenomas,1-3 and around 70% of all adrenocortical tumors are hormonally functional.1 Rarely, adrenal cortical tumors are bilateral. Bilateral diffuse adrenal cortical hyperplasia has been sporadically reported in cats. The stimulus for hyperplasia is unknown, but it is thought to be caused by an unidentified zona glomerulosa stimulatory factor or disturbances to the gonadal-pituitary axis.4
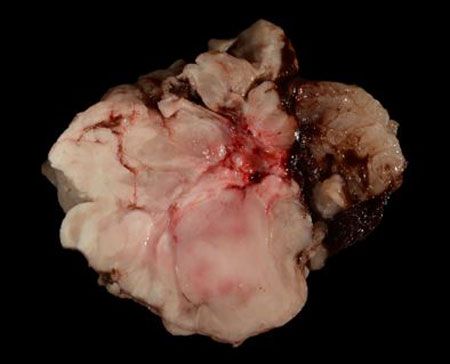
Figure 2: Gross image of the adrenocortical carcinoma diagnosed in this case. Note that no normal adrenal gland is identifiable.Differentiating adrenocortical adenomas and carcinomas can be challenging and requires correlating the clinical picture with the gross and histologic examination of the entire adrenal gland. Grossly, adenomas are usually smaller (less than 2 cm in dogs,5 with no size criteria reported in cats), well-demarcated, tan to yellow (similar to the normal cortex) and may be surrounded by compressed remaining cortex (Figure 1). In contrast, carcinomas are typically larger; have multifocal red, friable areas of hemorrhage and necrosis; and can invade into surrounding tissues, the vena cava or phrenicoabdominal vein or metastasize to distant sites (Figure 2).5 With hormonally functional tumors, there is often atrophy of the contralateral adrenal cortex.
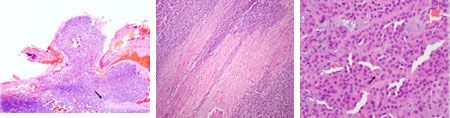
Figure 3 (left): The adrenocortical carcinoma diagnosed in this case displays invasion of the neoplastic cells through the adrenal capsule. Note disruption of the capsule (arrow) and extension of neoplastic cells through the capsule (hematoxylin-eosin; 40X). Figure 4 (center): The adrenocortical carcinoma diagnosed in this case displays multiple linear hypereosinophilic areas of coagulative necrosis (hematoxylin-eosin;100X). Figure 5 (right): The adrenocortical carcinoma diagnosed in this case displays mild to moderately pleomorphic polygonal cells with loss of cytoplasmic vacuoles and a central mitotic figure (arrow). Note the scattered large nuclei (karyomegaly) (hematoxylin-eosin; 400X).Histologically (Figures 3-5), adrenocortical tumors are composed of polygonal cells in cords, trabeculae or nests separated by minimal fibrovascular stroma. Adenomas are usually well-differentiated and surrounded by a fibrous connective tissue capsule or a thin rim of compressed cortex. Neoplastic cells usually have vacuolated light eosinophilic cytoplasm. Carcinomas can be well-differentiated, but they tend to be more pleomorphic with loss of cytoplasmic vacuoles, varying growth patterns and invasion through the capsule or into vessels. Both carcinomas and larger adenomas can have areas of hemorrhage and necrosis.5
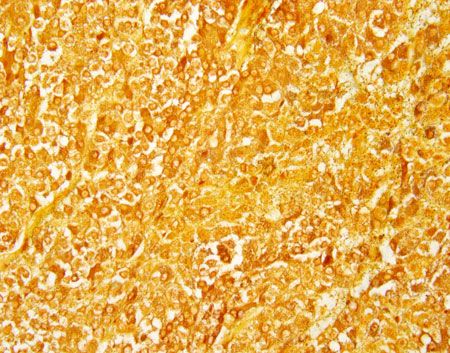
Figure 6: A feline adrenal medullary pheochromocytoma. Neoplastic cells have abundant cytoplasmic argyrophilic granules (Churukian-Schenk; 400X).Medullary pheochromocytomas are the other primary tumor of the adrenal gland and have been rarely reported in cats.1 Larger pheochromocytomas, which may resemble adrenocortical carcinomas, can be differentiated by the presence of argyrophilic granules, which are highlighted with a Churukian-Schenk stain (Figure 6).
References
1. Daniel G, Mahony OM, Markovich KM, et al. Clinical findings, diagnostics and outcome in 33 cats with adrenal neoplasia (2002-2013). J Feline Med Surg 2016;18(2):77-84.
2. Briscoe K, Barrs VR, Foster DF, et al. Hyperaldosteronism and hyperprogesteronism in a cat. J Feline Med Surg 2009;11(9):758-762.
3. Chiaramonte D, Greco DS. Feline adrenal disorders. Clin Tech Small Anim Pract 2007;22(1):26-31.
4. Javadi S, Djajadiningrat-Laanen SC, Kooistra HS, et al. Primary hyperaldosteronism, a mediator of progressive renal disease in cats. Domestic Anim Endocrinol 2005;28:85-104.
5. Meuten DJ. Tumors of the endocrine glands. In: Tumors in Domestic Animals. 5th ed. Ames, Iowa: Wiley Blackwell, 2016;782-791.
MEDICAL ONCOLOGY PERSPECTIVE
Lark Walters, DVM
Several prognostic factors have been identified in cats with hyperaldosteronism. Female sex and prolonged anesthesia time (more than four hours) in cats undergoing surgery have both been associated with decreased survival times.1,2 While long-term survival can be achieved with surgery, options are lacking when surgery is not possible or if metastatic disease is present.3 Tyrosine kinase inhibitors, such as toceranib phosphate (Palladia-Zoetis), may be considered, but this is extrapolated from anecdotal use in dogs and no information on efficacy exists in cats. Radiation therapy has been successfully used to treat adrenal tumors in dogs, but this has not been reported in cats.4 Therefore, when surgery is not possible or is declined, there are no well-established chemotherapeutic or radiation therapy options for adrenal tumors in cats, and supportive care with medical management (potassium supplementation and anti-hypertensives) is the recommended treatment.
References
1. Daniel G, Mahony OM, Markovich JE, et al. Clinical findings, diagnostics, and outcome in 33 cats with adrenal neoplasia (2002-2013). J Fel Med Surg 2014;18:77-84.
2. Lo AJ, Holt DE, Brown DC, et al. Treatment of aldosterone-secreting adrenocortical tumors in cats by unilateral adrenalectomy: 10 cases (2002-2012). J Vet Intern Med 2014;28:137-143.
3. Withrow SJ, Page R, Vail DM. Tumors of the endocrine system. In: Small animal clinical oncology. 5th ed. St. Louis, Missouri: Elsevier, 2013.
4. Dolera M, Malfassi L, Pavesi S, et al. Volumetric-modulated arc stereotactic radiotherapy for canine adrenocortical tumours with vascular invasion. J Small Anim Pract 2016;12:710-717.