Crystalloids or colloids?
Vital information to help you decided which IV fluid is best in each patient.
Exactly how to make the decision to use a crystalloid or colloid fluid for the perioperative prevention or treatment of hypotension and hypovolemia is still undefined. What has become evident is that the negative effects of fluid overload increase postoperative morbidity and mortality regardless of fluid choice. Knowing the fundamental differences between and the potential benefits of the various types of crystalloids and colloids is essential to their selection as replacement therapy in surgical candidates.
PHOTO BY GREGORY KINDRED
This article reviews salient aspects for the selection of fluids for intravenous perioperative fluid therapy. Intravenous fluid solutions are broadly classified as crystalloids (salts in water) or colloids (noncrystalline substances consisting of large molecules diluted in a crystalloid). The capillary endothelium is freely permeable to water and dissolved salts (e.g. sodium, chloride) but is relatively impermeable to larger molecules (> 30,000 Da).
CRYSTALLOIDS
Sodium chloride
Normal saline solution (0.9% sodium chloride), often referred to as physiological saline solution, has a tonicity (osmotic pressure) similar to that of plasma (308 mOsm/L) but is actually a nonphysiological, chloride-rich, unbalanced salt solution that, when given intravenously in large volumes (> 30 ml/kg), produces metabolic acidosis, has the potential to impair renal blood flow, and predisposes the animal to postoperative vomiting.1-3
The perioperative administration of 0.9% sodium chloride produces a higher risk of morbidity and mortality and is more likely to produce adverse events in surgical patients than balanced crystalloid solutions.4
Balanced solutions
Physiological and balanced salt solutions possess electrolyte concentrations that have a tonicity similar to that of plasma (290 to 310 mOsm/L: isotonic) and help maintain physiological pH, respectively. Conventional crystalloid replacement solutions are designed to mimic plasma and contain molecules (lactate, acetate, gluconate; ≥ 24 to 28 mEq/L) that can be converted to bicarbonate5,6 in order to minimize changes in plasma pH (approximately 7.4).6
Balanced crystalloid solutions help maintain normal hydration, electrolyte concentrations, and acid-base balance during surgery; however, their ability to improve hemodynamics (cardiac filling pressures, arterial blood pressure, cardiac output) and tissue perfusion (microcirculatory blood flow) is highly variable and generally transient.7,8 Keep in mind that all isotonic crystalloids produce increases in blood flow (cardiac output) and small decreases in arterial blood pressure because of a decrease in plasma viscosity.9,10
Hypotonic and hypertonic crystalloids add water to or extract water from the extravascular fluid compartment, respectively.
Hypotonic solutions. Examples of minimally hypotonic and isotonic replacement solutions administered to treat hypovolemia and hypotension include lactated Ringer's solution (Abbott Animal Health), Plasma-Lyte A (Baxter), and Normosol R (Hospira, Inc.) (Table 1).

Table 1: Crystalloid solutions*
Hypotonic and isotonic crystalloid solutions are poor plasma (volume) expanders. Studies in anesthetized dogs administered an intravenous crystalloid at rates equal to or exceeding 1 ml/kg/min (> 60 ml/kg/hr) suggest that 40% to 75% of the infused volume is retained in the vascular compartment immediately after infusion but that this value decreases to less than 25% within 20 to 30 minutes.8,11
Hypertonic solutions. Hypertonic crystalloid solutions are considered to be plasma expanders because their tonicity causes water to move from interstitial and intracellular sites into the intravascular compartment, thereby increasing plasma volume.12 Hypertonic saline solution (5% to 7.5% sodium chloride) is a unique crystalloid demonstrated to rapidly expand plasma volume. It produces acute beneficial hemodynamic, immunologic, and microcirculatory effects when administered in comparatively low total volumes (3 to 4 ml/kg), thereby decreasing the potential for adverse effects (see "Sodium chloride" above) and volume overload.12,13
Hypertonic saline solution has also been advocated as therapy for traumatic brain injury.4 However, questions about the effects of hypertonic saline solutions on mortality in people remain unresolved and require further investigation.12,13
COLLOIDS
Colloids are more effective than crystalloids for correcting intravascular volume deficits and for improving systemic and microcirculatory blood flow, even if the crystalloid is rapidly infused.14-17 Colloid solutions contain molecules, such as albumin, that produce a colloid osmotic (oncotic) pressure (COP) and, thus, help retain fluid within the blood vessel. Colloidal solutions can be either natural (e.g. albumin) or synthetic (e.g. dextran, hydroxyethyl starch) and are frequently categorized based on their molecular weight.
The benefits of COP
Compared with crystalloids, colloids are excellent replacement fluids because of their ability to produce COP.14,18,19 The capacity of a colloid to expand the blood volume, although variable, is directly related to its ability to increase COP (Table 2).19,20 The COP is related to the number of active particles available in solution.

Table 2: Hydroxyethyl starch solutions
Increases in plasma COP help retain fluid in the vascular compartment and, under the right circumstances, promote fluid movement from the interstitial fluid compartment into the vascular fluid compartment (autotransfusion).
Colloids produce a greater and more sustained increase in vascular volume than crystalloids do. Notably, 70% to 80% of a crystalloid solution (COP = 0 mm Hg) is distributed into the interstitial fluid compartment within 15 to 30 minutes of its administration, while colloidal solutions produce effects that are variably sustained for hours after their administration. The colloid effects depend on their colligative and chemical properties and the integrity of the vessel wall.8,21
A look at synthetic colloids
The synthetic colloids hetastarch (e.g. Hespan—Abbott Animal Health; Hextend—Hospira, Inc.) and tetrastarch (e.g. VetStarch—Abbott Animal Health; Voluven—Hospira, Inc.) are hydroxyethyl starch solutions that vary in molecular weight, molar substitution (number of hydroxyethyl residues per glucose subunits), and the pattern of substitution of hydroxyethyl residues on glucose subunits (C2/C6 ratio).20 These properties determine the magnitude and duration of their volume-expanding effects (Table 2). Higher molecular weight, molar substitution, and C2/C6 ratios prolong plasma retention and the duration of volume-expanding effect.
Hydroxyethyl starch solutions with lower molecular weight and molar substitution have shorter half-lives and produce fewer side effects, yet retain clinically important volume-expanding efficacy because they contain more molecules per milliliter (produce a greater COP) than a product with a high molecular weight.
Tetrastarch (4 hydroxyethyl residues per 10 glucose subunits; 4/10 = 0.4) in 0.9% sodium chloride has a mean molecular weight of 130 kDa and molar substitution of 0.4 (130/0.4). It produces immediate and sustained increases in hemodynamics with comparatively lower risk of fluid overload, coagulation derangements (factor VIII, platelet function), renal impairment, or tissue accumulation than hetastrach solutions, provided that dosing guidelines are followed.20,22-28 Tissue oxygenation is improved29 by improving microcirculatory blood flow.14,17
Note that in contrast to other commercially available hydroxyethyl starch solutions, tetrastarch (VetStarch) is approved for both the treatment and prevention of hypovolemia. Total daily doses of VetStarch should not exceed 50 ml/kg/day for most animals and 15 ml/kg/day for animals with prolonged bleeding times (prothrombin time >13 sec; partial thromboplastin time > 24 sec), reduced platelet numbers (< 50,000/ml), or elevated creatinine concentrations (> 1.8 mg/dl [157 mmol/L]).
Clinical experience after administration of tetrastarch (VetStarch) doses, ranging from 20 to 50 ml/kg/day, in critically ill dogs suggests good to excellent volume replacement without discernable adverse effects (Table 3). Focused clinical trials determining efficacy and effects on coagulation and renal function in critically ill animals are still needed.
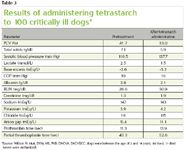
Table 3: Results of administering tetrastarch to 100 critically ill dogs*
Precautions
Caution is always advised when administering a colloid in dehydrated animals or animals with evidence of coagulopathy, head trauma, or impaired renal function (elevated BUN or creatinine concentrations).30-32 Adverse effects and toxicities caused by colloidal solutions are dose-dependent, and guidelines for dosing should be strictly followed. Notably, human hospital trials suggest that tetrastarch (HES 130/0.4)-associated renal impairment is low and, compared with other hydroxyethyl starches, is most likely to occur in animals who have failed to receive sufficient water or who remain hypotensive or when recommended maximum daily doses are exceeded.31-33
CONCLUSION
Provided that arterial blood pressure remains within normal limits, isotonic balanced crystalloids solutions are adequate therapy for perioperative treatment caused by insensible fluid loss (e.g. evaporation from open body cavities) and anesthesia-associated systemic vasodilatation (relative hypovolemia). Colloids produce more immediate and sustained beneficial hemodynamic (macrocirculatory and microcirculatory) effects and are effective replacement therapy for perioperative hypotension and hypovolemia (relative or absolute).
The packed cell volume should be monitored during fluid therapy and maintained at values greater than 20%. Current knowledge and veterinary clinical guidelines for perioperative fluid management are primarily based on opinion, nonclinical animal research, and human clinical trials. Human clinical trials recommend not to administer colloidal solutions in any subject with evidence of head trauma or increased risk of acute kidney injury.
ACKNOWLEDGMENTS
The author thanks Dava Cazzolli, DVM, DACVECC; Jaime Chandler, DVM, DACVECC; Michelle Albino, LVT, CVT (Anesth); and Yukie Ueyama, DVM, for their assistance in abstracting data.
William W. Muir, DVM, MS, PhD, DACVA, DACVECC
VCPCS
338 W. 7th Ave.
Columbus, OH 43201
For this article's reference citations, go to dvm360.com/FluidTherapy2Refs.