On the Forefront: Cardiac cellular transplantation: A future treatment for heart disease?
CCT aims to replace lost cardiomyocytes with progenitor cells capable of growing into new cardiac-like tissue.
Over the years, the diagnosis and treatment of cardiac disease in dogs and cats have improved through the development of new technology (echocardiography), the introduction of new drugs (ACE inhibitors, calcium sensitizers), and a growing understanding of the cause and pathophysiology of disease. Yet despite these advances, the morbidity and mortality associated with cardiac disease remain high.
In human medicine, the most effective medications are those that alter the underlying pathophysiology of disease, usually by correcting the abnormal activity of neurohormonal mechanisms such as the renin-angiotensin or sympathetic nervous system. However, even these medications cannot replace cardiac muscle cells (cardiomyocytes) that are irretrievably lost to the underlying disease. Unlike other types of cells, cardiomyocytes are terminally differentiated and are incapable of returning to the cell cycle. Progressive loss of viable cardiomyocytes, a feature of heart disease in both people and animals, leads to further stress on the remaining tissue, sustained activation of detrimental neurohormonal activity, and progressive loss of global cardiac function.
USING CARDIAC CELLULAR TRANSPLANTATION TO TREAT CARDIAC DISEASE
Cardiac cellular transplantation (CCT), a novel heart disease treatment, aims to replace lost cardiomyocytes with progenitor cells capable of growing into new cardiac-like tissue. Many organ systems can regenerate new tissue; the liver, skin, bone marrow, and skeletal muscle all contain populations of precursor cells capable of rebuilding damaged tissue. For example, skeletal muscle contains myoblast (satellite) cells that reside in the periphery of the muscle fiber. These myoblasts are recruited to areas of muscle injury to engraft and differentiate into new functional skeletal muscle. Other examples of these adult stem cells include bone marrow and endothelial progenitor cells. Using stem cells derived from adult tissue instead of those from embryonic tissue has several advantages, the most important being the avoidance of the ethical dilemma surrounding embryonic stem cell use. Also, using adult stem cells from a patient's own (autologous) tissue removes the concern over transplant rejection.
Atherosclerosis-related myocardial infarctions
CCT has been successfully performed in people with atherosclerosis-related myocardial infarctions. During this procedure, aliquots of stem cells are directly injected into the heart muscle with a syringe and needle at the time of coronary artery bypass grafting. The results from early trials in people as well as from a growing amount of animal data suggest that, when implanted, a variety of adult stem cells are able to survive and differentiate into cardiac-like muscle tissue, rebuilding the heart one cell at a time.
Canine dilated cardiomyopathy
Unlike ischemic disease in people, which is primarily a regional phenomenon, canine dilated cardiomyopathy is a disease of global ventricular proportion, and local injections of cells during surgery would be a relatively inefficient way to distribute stem cells throughout the affected left ventricle. In 2000, researchers reported a technique in which stem cells were injected into the coronary circulation where they were entrapped within the capillary beds and subsequently disseminated into the surrounding tissue.1 The advantage of this technique is a more uniform and global distribution of cells.
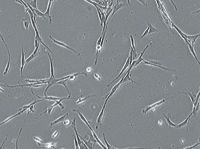
Figure 1 : Phase contrast microscopy of canine myoblasts isolated through cell culture displaying stellate-shaped myoblasts.
Our research at the University of Illinois College of Veterinary Medicine has demonstrated the feasibility of autologous CCT in dogs by using skeletal muscle myoblasts delivered through a coronary artery injection. We are investigating this new technology with the future hope of treating dogs that have dilated cardiomyopathy. With our procedure, skeletal muscle biopsy samples are obtained from the quadriceps muscle and are used to harvest myoblasts through a series of filtration and incubation steps. Over several weeks, the myoblasts are expanded in cell culture (Figure 1) and then labeled with a fluorescent dye so they are identifiable after transplant (Figure 2). The cells are then injected back into the donor dog by using a fluoroscopically guided catheter placed into the left coronary artery.

Figure 2 : A fluorescent microscopic image of labeled myoblasts within the left ventricular tissue of a dog that received one million myoblasts through coronary artery injection. The transplanted cells are identified by their blue color against the nonfluorescent background of native myocardial cells.
This catheter-based injection technique only requires access to the femoral artery and does not require a thoracotomy. We have injected up to five million myoblasts into the hearts of normal dogs and have preliminarily established the feasibility and safety of this technique. We have also examined the efficacy of myoblast entrapment within the myocardial capillary bed and documented the survival of the transplanted cells up to seven days after the transplant.
MORE RESEARCH IS NEEDED
Stem cell research has the potential to dramatically change the way many cardiac and noncardiac diseases and injuries are treated, including diabetes, spinal cord injury, muscular dystrophy, and bone and cartilage diseases. CCT has garnered an enormous amount of attention in the scientific community, and considerable resources and effort are being applied to its study. Despite early promise, much work is still needed to ascertain the long-term efficacy of CCT and to refine transplantation and cell culture techniques. The clinical evaluation of CCT is just beginning, and we need a better understanding of the fundamental biologic processes that govern the behavior of these cells. For instance, little is known about the actual process of stem cell differentiation and the specific cellular, genetic, and molecular events that lead to successful maturation and incorporation into the surrounding tissue, nor is it known which adult cell population (e.g. myoblasts vs. bone marrow mesenchymal cells) is the ideal population for transplant. Future investigations should help answer many of these questions and determine whether the promise of CCT can be realized.
ACKNOWLEDGMENTS
The author would like to acknowledge co-investigators Tom Eurell, DVM, PhD, DABT, and Valentina Merola, DVM.
REFERENCE
1. Suzuki K, Brand NJ, Smolenski RT, et al. Development of a novel method for cell transplantation through the coronary artery. Circulation 2000;19(suppl 3):III359-III364.
The information and photographs for "On the Forefront" were provided by Mark A. Oyama, DVM, DACVIM (cardiology), Department of Veterinary Clinical Medicine, College of Veterinary Medicine, University of Illinois, Urbana, IL 61802.
Podcast CE: Canine cardiology: the practical guide to the mitral valve patient
July 19th 2023Learn about the prevalence of myxomatous mitral valve disease, guidelines for staging heart disease, proactive diagnostic workup, the importance of spironolactone and aldosterone blocking, and the benefits of combination therapy for improved outcomes in canine patients
Listen