On the Forefront: Extended-release liposomal opioids for treating pain in dogs
Adequate analgesic treatment of both acute perioperative pain and chronic pain in dogs is recognized by the veterinary profession and the general public as imperative to appropriate and humane medical care.
Adequate analgesic treatment of both acute perioperative pain and chronic pain in dogs is recognized by the veterinary profession and the general public as imperative to appropriate and humane medical care.1-3 Because of modern veterinary medicine's more sophisticated invasive surgical procedures (e.g. thoracotomy), moderate to severe perioperative pain can be expected to last at least 48 to 72 hours after surgery. Also, because of improved veterinary preventive care, dogs have an increased life expectancy, and with increasing age comes painful age-related conditions such as osteoarthritis.
Wanted: A perfect analgesic
Of the available analgesic regimens in veterinary medicine, opioids remain the gold standard for moderate to severe pain in dogs.4 Most injectable opioids require dosing every three to six hours (or more often) to maintain therapeutic serum concentrations. Currently available oral formulations of opioids, such as morphine sulfate controlled-release tablets (MS Contin—Purdue Frederick), have a significant first-pass effect in dogs, rendering the actual analgesic efficacy of this formulation questionable.5
Transdermal fentanyl patches have gained popularity in veterinary medicine, with effective serum fentanyl concentrations being achieved within 24 hours and maintained for about 72 hours after patch application.6 When a dog is sent home with a fentanyl patch, however, there is a risk of access to the patch by caretakers or children in the household.7,8 In addition, the patch's effectiveness varies depending on how well it sticks to the dog's skin, and the dog can become profoundly sedated if it ingests the patch.
No currently available formulations of opioids for use in dogs or other species meet all the desirable characteristics of adequate analgesia: effectiveness, predictable therapeutic serum concentrations, a convenient dosing interval, minimal side effects, and no risk of diverting the drug to a person or accidental ingestion by the pet or by children.
Liposomal opioid formulations: A possible solution?
Our group, which consists of a clinical anesthesiologist (L.J. Smith) and a laboratory animal veterinarian (L.A. Krugner-Higby), at the University of Wisconsin's School of Veterinary Medicine, in collaboration with chemists at the University of Wisconsin's School of Pharmacy, has been developing novel liposome-encapsulated formulations of both oxymorphone and hydromorphone. These formulations are synthesized such that the opioid is surrounded by onion-skin–like layers of lipid bilayer membranes, with opioid sandwiched between layers (Figure 1). The drug is released by both slow leakage across the lipid membranes, depending on the drug's lipid solubility, and by membrane degradation by local tissue macrophages after the drug is injected into the animal.
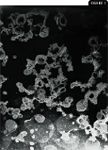
Figure 1. A scanning electron micrograph of the type of liposomes being formulated in our lab. The liposomal membranes were stained with 2% phosphotungstic acid in this preparation to highlight the membrane structure.
These formulations show promise as extended-release preparations of opioids, providing therapeutic drug concentrations for up to seven days after a single subcutaneous injection.9 Our initial work involved the synthesis of liposomal oxymorphone by using egg phosphatidylcholine, with about 80% recapture of the oxymorphone hydrochloride base.9 This simple egg phosphatidylcholine liposome had in vitro release characteristics that suggested a potential analgesic duration of five to seven days.9
We have since developed liposomal oxymorphone and hydromorphone with a more stable liposomal membrane made from both cholesterol and dipalmitoylphosphatidylcholine (DPPC) that has a release time of over 13 days (Figure 2). Both egg phosphatidylcholine and DPPC-cholesterol liposomal opioid formulations not only have predictable release times but also offer potential for a convenient dosing interval of once every one or two weeks; a subcutaneous route of administration by the veterinarian, making access by the pet or by human caretakers impossible; and reduced side effects because of the slow, steady release of drug without a bolus effect.
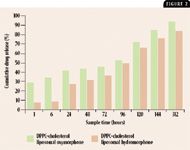
Figure 2. In vitro characteristics of DPPC-cholesterol liposomal oxymorphone and hydromorphone drug release over time.
Since developing liposomal oxymorphone and hydromorphone in the laboratory, we have tested their efficacy and safety in rats, mice, dogs, and, recently, nonhuman primates. Much of this data is preliminary, but we have recently gained funding from the Morris Animal Foundation and from the National Institutes of Health that allows us to investigate the potential clinical application of these drug formulations.
Thus far, we have shown that one subcutaneous injection of liposomal oxymorphone provides effective analgesia for seven days in a rat model of neuropathic pain9 and in a rat and mouse model of postoperative visceral pain.10,11 We have also shown that a single dose of liposomal oxymorphone provided therapeutic serum concentrations of drug for three days in healthy dogs.12 Work with liposomal hydromorphone in rats has recently been completed (Smith LJ, et al, School of Veterinary Medicine, University of Wisconsin, Madison, WI: Unpublished data, 2005). These studies were all performed with egg phosphatidylcholine liposomes; studies with cholesterol-containing DPPC liposome-encapsulated oxymorphone or hydromorphone in animals will be conducted to evaluate the duration of therapeutic serum concentrations.
Currently, our laboratory is focusing on pharmacokinetic studies in dogs, rats, and nonhuman primates to better describe the biologic behavior of liposomal oxymorphone and hydromorphone. We are also testing analgesic thresholds after administering these drugs to healthy, nonpainful animals. We plan to study the side effects of these drug formulations by evaluating mental status, heart rate, and arterial blood gases.
The next steps
Liposomal oxymorphone and hydromorphone are not yet commercially available. We anticipate that in the next several years we will be testing the analgesic efficacy of liposomal opioids in client-owned dogs with postoperative pain and dogs with chronically painful conditions such as osteoarthritis or cancer. In the future, we may investigate the effects of liposomal opioids given epidurally.
Much potential exists in the clinical application of liposomal opioids for many veterinary and laboratory animal species. The extended dosing interval and reduced potential for access make them ideal for use in large laboratory animal colonies.
Questions concerning current or future studies with liposomal oxymorphone or liposomal hydromorphone can be directed to Dr. Lesley J. Smith at the Department of Surgical Sciences, School of Veterinary Medicine, University of Wisconsin, 2015 Linden Drive, Madison WI 53706; or call (608) 263-9808, fax (608) 263-7930, or e-mail smithl@svm.vetmed.wisc.edu
REFERENCES
1. Hellyer PW. Treatment of pain in dogs and cats. J Am Vet Med Assoc 2002;221:212-215.
2. Lascelles BDX, Main DCJ. Surgical trauma and chronically painful conditions–within our comfort level but beyond theirs? J Am Vet Med Assoc 2002;221:215-222.
3. Kyles AE, Ruslander D. Chronic pain: osteoarthritis and cancer. Semin Vet Med Surg (Small Animal) 1997;12:122-132.
4. Haskins SC. Advantages and guidelines for using agonist opioid analgesics. Vet Clin North Am Small Anim Pract 1992;22:360-362.
5. Dohoo S. Steady-state pharmacokinetics of oral sustained-release morphine sulphate in dogs. J Vet Pharmacol Ther 1997;20:129-133.
6. Kyles AE, Papich M, Hardie EM. Disposition of transdermally administered fentanyl in dogs. Am J Vet Res 1996;57:715-719.
7. Hardwick WE Jr, King WD, Palmisano PA. Respiratory depression in a child unintentionally exposed to transdermal fentanyl patch. South Med J 1997;90:962-964.
8. Purucker M, Swann W. Potential for duragesic patch abuse. Ann Emerg Med 2000;35:314.
9. Smith LJ, Krugner-Higby L, Clark M, et al. A single dose of liposome-encapsulated oxymorphone or morphine provides long-term analgesia in an animal model of neuropathic pain. Comp Med 2003;53:280-287.
10. Krugner-Higby L, Smith LJ, Clark M, et al. Liposome-encapsulated oxymorphone hydrochloride provides prolonged relief of postsurgical visceral pain in rats. Comp Med 2003;53:270-279.
11. Clark MD, Krugner-Higby L, Smith LJ, et al. Evaluation of liposome-encapsulated oxymorphone hydrochloride in mice after splenectomy. Comp Med 2004;54:558-563.
12. Smith LJ, Krugner-Higby L, Trepanier LA, et al. Sedative effects and serum drug concentrations of oxymorphone and metabolites after subcutaneous administration of a liposome-encapsulated formulation in dogs. J Vet Pharmacol Ther 2004;27:369-372.
The information for "On the Forefront" was provided by Lesley J. Smith, DVM, DACVA, Department of Surgical Sciences, School of Veterinary Medicine, University of Wisconsin, Madison, WI 53706.