Scrotal castration versus prescrotal castration in dogs
Is the scrotal castration technique as safe and efficient as the commonly taught prescrotal technique? The results of this study might surprise you.
For many years, the prescrotal technique has been taught as the only acceptable method of canine castration.1 However, scrotal castration has gained popularity in recent years as a safe alternative to the prescrotal technique. First described in 1974,2 this technique may offer the advantage of reducing surgical time while not increasing complication rates over the traditional prescrotal approach.
The scrotal technique has been described as an accepted method for pediatric canine castrations.1 It is becoming widely accepted for adult canine castrations by veterinary surgeons in high-volume spay-neuter clinics, which often have limited resources and many animals to sterilize.3 Procedures that reduce anesthetic time and expedite the surgical procedure by even a few minutes can be of tremendous benefit to these programs.
Although numerous clinics perform the scrotal technique, to our knowledge, there is no published research documenting its complication rate or comparing complications between the scrotal and prescrotal techniques. Our study was conducted to compare complication rates and surgical efficiency between the two castration techniques in male dogs more than 6 months old. We wanted to evaluate the hypothesis that there are no differences in complication rates between the two techniques.
COMPLICATIONS
Orchiectomy, like all surgeries, carries risks of complication. While there is a perception that scrotal castration in adult dogs is more prone to complications than prescrotal castration is, limited data are available comparing complication rates of scrotal and prescrotal canine castrations. Data are difficult to obtain because complications and degree of detail of records vary by practitioner.4,5 Additionally, some minor complications occur at home and may go unnoticed or unreported by owners. Complication rates after prescrotal castrations have been reported to range from 0% to 32%, with the incidence of complications often considered to be lower in younger patients.4,6
Complications of both prescrotal and scrotal techniques include dehiscence, scrotal swelling, hemorrhage, subcutaneous bruising, scrotal hematoma and self-trauma to the surgical site. Dogs with minor complications may need no intervention, while others may require veterinary care. In one study of 218 animals, seven dogs and two cats developed scrotal hematoma after castration.3,7 Dogs with severe scrotal hematoma may experience necrosis of the scrotal skin, necessitating a scrotal ablation.7
In the past, scrotal castrations have been discouraged because male dogs are considered to be scrotal conscious.8 The accepted thought has been that disturbing the scrotal skin will cause excessive self-mutilation by the patient, most likely because of irritation caused by skin sutures.3 For this reason, several studies have discouraged clipping or prepping the scrotum at all and have recommended draping the scrotum out of the surgical field.8 The potential for self-mutilation has been given as the reason to avoid performing scrotal castrations, despite the fact that there is no reported scientific evidence supporting this conclusion.3
Recently, advances have been made in surgical and diagnostic procedures, especially in human medicine, toward less invasive techniques. These advances have led to reduced morbidity and wound contamination as well as less pain and shorter patient recovery periods.9 While this practice is developing at a slower rate in veterinary medicine, there are ongoing efforts to make common procedures less invasive. The scrotal technique, although not well-documented, is considered by many veterinarians working at high-quality high-volume spay-and-neuter clinics to be quicker and less invasive than the traditional prescrotal approach.3
METHODOLOGY
Dogs for this study were selected from five shelters serviced by the Mississippi State University (MSU) mobile surgical unit and from all dogs presented for castration to Humane Alliance (HA) in Asheville, North Carolina. All dogs were healthy males more than 6 months old. Dogs with signs of disease or cryptorchidism were excluded from the study. The dogs were randomly allocated by a coin toss into two treatment groups, scrotal castration and prescrotal castration. Both groups of dogs were tattooed after surgery to identify the procedure performed.
Time efficiency was recorded for the procedures done by the MSU surgeons. Efficiency was measured in minutes of surgical time, starting when the surgeon made the incision and concluding after the last suture was placed. The duration of each surgery was recorded by a veterinary assistant present in the surgery suite. No data on the duration of surgery were recorded at HA.
Surgical preparation
Dogs were anesthetized with butorphanol (0.35 mg/kg), ketamine (3.5 mg/kg), and dexmedetomidine (17.5 µg/kg) given as an intravenous mixture. Before surgery, each dog was given a subcutaneous injection of carprofen (4.4 mg/kg) for pain control.
For both procedures, the surgical area, including the scrotum and prescrotal area, was clipped and prepared with chlorhexidine scrub, and the surgical area was covered with a clean, chlorhexidine-soaked surgical sponge. The dog was then moved to the surgical suite and placed in dorsal recumbency. The clean surgical sponge was removed, and the surgical site was aseptically draped. There were no differences in surgical prep between the two techniques (Figures 1A & 1B).
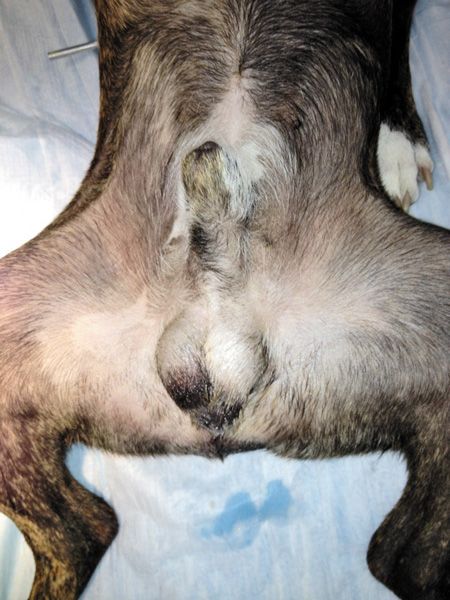
1A. A dog aseptically prepared for scrotal castration. There were no differences in surgical preparation between the two techniques.
1B. A dog prepped for prescrotal castration.
Prescrotal incision technique
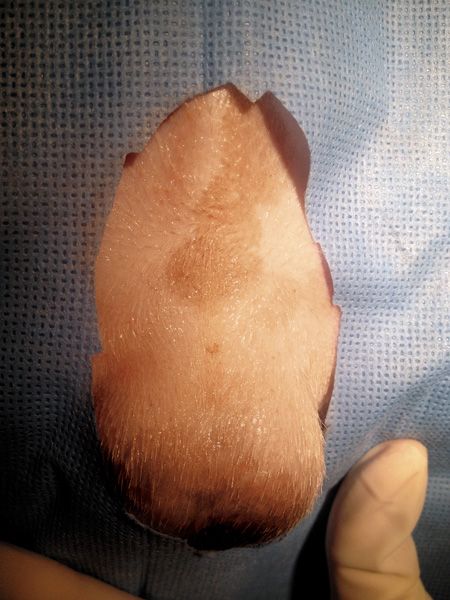
For those dogs undergoing the prescrotal incision technique, a No. 15 scalpel blade on a No. 2 Bard-Parker handle was used to incise the prescrotal skin. The incision was made just cranial to the scrotum and continued cranially 2 to 5 cm, depending on the dog's size, until the incision was of sufficient length to allow the testicles to be exteriorized (Figure 2). The parietal tunic was left intact.
2. The prescrotal incision was made just cranial to the scrotum and continued cranially 2 to 5 cm, depending on the dog's size, until the incision was of sufficient length to allow the testicles to be exteriorized.
The first testicle was delivered through the prescrotal incision; fascia was stripped from the spermatic cord to allow the testicle and spermatic cord to be fully exteriorized for a closed castration technique. Two curved Kelly hemostats were used to crush the tissues of the spermatic cord proximal to the testicle. The spermatic cord was transected distal to the second hemostat with a No. 15 scalpel blade. The most proximal hemostat was removed, and ligature of 2-0 polyglactin 910 (Vicryl-Ethicon) suture was secured with a Miller's knot in the area previously crushed by the hemostat.
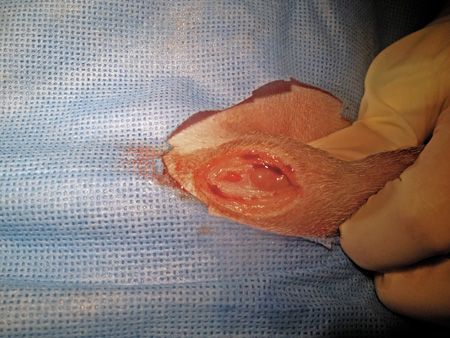
The remaining hemostat was subsequently removed, and the remainder of the spermatic cord was placed back into the incision after checking for hemorrhage. The procedure was repeated for the second testicle. The incision was closed with 2-0 polyglactin 910 suture in an interrupted intradermal pattern (Figure 3).
3. The prescrotal incision was closed with 2-0 polyglactin 910 suture in an interrupted intradermal pattern.
Scrotal technique
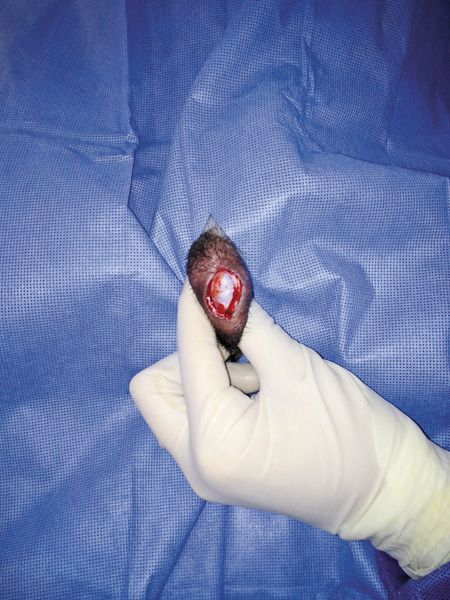
4. For the scrotal technique, the first testicle was delivered through a scrotal incision near the median raphe.
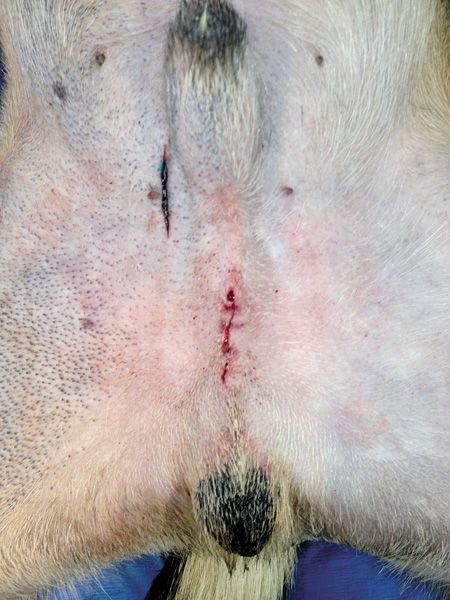
For the scrotal technique, a No. 15 scalpel blade on a No. 2 Bard-Parker handle was used to make a 2- to 5-cm incision in the scrotum (Figure 4). The first testicle was delivered through a scrotal incision near the median raphe, and a closed castration was performed as described for the prescrotal technique. The procedure was repeated for the second testicle using the same incision. A single subcutaneous suture was placed in the scrotal incision by using 2-0 polyglactin 910 suture (Figure 5).
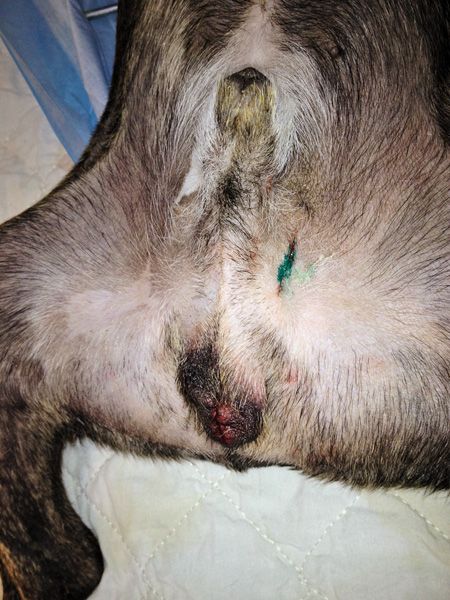
5. A single subcutaneous suture was placed in the scrotal incision by using 2-0 polyglactin 910 suture. Both groups of dogs were tattooed to identify the procedure performed.
Postoperative monitoring
The dogs were placed in a cage or run and monitored during recovery. Dogs were ultimately returned to the shelter environment within two hours. Privately owned dogs were returned to their owners about 24 hours after surgery.
Dogs treated at MSU were monitored by shelter employees, while dogs treated at HA were monitored by individual owners. Whenever possible, the same individual assessed multiple dogs. All observers were given verbal and written instructions concerning proper observation and documentation of complications on a provided questionnaire (seeCastration postoperative assessment form). Complications were defined as the presence and absence of hemorrhage (blood from the incision site), pain (vocalization on palpation of the incision site), self-trauma (licking, chewing or scratching at the incision), and swelling of the incision site or scrotum. Swelling was evaluated two, four, six, 24, 48 and 72 hours after surgery.
Results
Four hundred thirty-seven dogs met the inclusion criteria for this study. The average weight of the dogs included was 37.5 lb (17 kg) and ranged from 6.6 to 132.3 lb (3 to 60 kg). The prescrotal approach was performed on 206 dogs, and 231 were castrated by using the scrotal approach. Surgeries were performed by nine licensed veterinarians. All veterinarians were proficient in high-quality high-volume spay-neuter techniques and had a minimum of four years of experience. No complications were noted during the surgical procedures.
For statistical purposes, the frequencies of complications were categorized by method and location (Table 1). The mean weights for dogs with or without complications are also presented in Table 1. The results of the multivariable logistic regression analysis are listed in Table 2. (SeeData assessment.)
It is interesting to note that 54 dogs (prescrotal = 34; scrotal = 20) were recorded as inflicting self-trauma through biting, licking or chewing their incisions (Table 1). The odds of self-trauma were 1.96 times greater (P = 0.04) in dogs undergoing the prescrotal method than in those castrated by the scrotal method when adjusted for state and weight (Table 2).
The odds of hemorrhage (26.45), pain (8.11) or self-trauma (14.66) were significantly greater (P < 0.01), when adjusted for method and weight, in dogs castrated at MSU than in those castrated at HA. The odds of hemorrhage, when adjusted for method and state, were 1.04 times greater (P = 0.02) for each 1-kg increase in weight of the dog.
The odds of swelling from two to six hours after surgery were significantly greater (P < 0.01), when adjusted for method and weight, in dogs castrated at MSU; however, the odds of swelling from 24 to 72 hours were significantly less (P ≤ 0.04) in dogs castrated at MSU.
Overall, dogs with prescrotal incisions had significantly higher incidence of self-trauma. These data are noteworthy considering the perception of scrotal consciousness in dogs and do not support the concern that a scrotal approach may increase the incidence of self-trauma.8 Larger dogs had greater odds for hemorrhage, but that was found to be independent of method.
The length of surgery was recorded for cases at MSU. A significant difference (P < 0.01) was recorded between the two procedures, with the average surgical time for the scrotal approach being 5.1 minutes and the average surgical time for the scrotal approach being 3.6 minutes, which is about a 30% reduction in surgical time (Table 3). The difference in surgical time by surgical approach was consistent between the two MSU surgeons.
DISCUSSION
Canine castration is one of the most common procedures performed in veterinary medicine, and the prescrotal surgical approach has traditionally been the most commonly taught method.1,6 The emergence of high-quality high-volume spay-neuter organizations has increased the need for more efficient techniques. Consideration should be given to other possible approaches that may be as effective, safe and efficient as the long-accepted prescrotal castration. To our knowledge, this study was the first designed to evaluate the differences in complication rate and time efficiency between scrotal and prescrotal canine castration.
In this study, the complication rates of the prescrotal and scrotal techniques were similar, but the scrotal approach was faster and had lowered incidence of self-trauma.
In future studies, efforts should be made to eliminate or further minimize interobserver variability (see Study limitations). While swelling was tracked out to 72 hours after surgery, pain, self-trauma, and hemorrhage were recorded only in the 24 hours immediately following the patient's recovery from anesthesia. It may be useful to follow the incidence of pain, self-trauma, hemorrhage, incisional discharge and infection rates out to at least seven days.
CONCLUSION
Scrotal castration was comparable with traditional prescrotal castration in terms of incidence of most postoperative complications. However, the scrotal method was associated with less self-trauma. Scrotal castration also offered an approximately 30% faster surgery time. Either surgical method may be safely and effectively performed in high-quality high-volume spay-neuter clinics.
Kimberly Woodruff, DVM, MS
Philip A. Bushby, DVM, MS, DACVS
Department of Clinical Sciences
College of Veterinary Medicine
Mississippi State University
Mississippi State, MS 39762
Karla Rigdon-Brestle, DVM
Humane Alliance
25 Heritage Drive
Asheville, NC 28806
Robert Wills, DVM, PhD, DACVPM
Carla Huston, DVM, MS, DACVPM
Department of Pathobiology and Population Medicine
College of Veterinary Medicine
Mississippi State University
Mississippi State, MS 39762
References
1. Fossum TW. Surgery of the reproductive and genital systems. In: Small animal surgery. 2nd ed. Philadelphia: Elsevier, 2002;619-620.
2. Archibald J. Male genital system. In: Canine surgery. 2nd ed. Santa Barbara, California: American Veterinary Publications, 1974;727-729.
3. Miller L, Zawistowski S. Efficient spay neuter techniques. In: Shelter medicine for veterinarians and staff. 2nd ed. Ames, Iowa: Wiley-Blackwell, 2013;635-637.
4. Pollari FL, Bonnett BN, Bamsey SC, et al. Postoperative complications of elective surgeries in dogs and cats determined by examining electronic and paper medical records. J Am Vet Med Assoc 1996;208:1882-1886.
5. Pollari FL, Bonnett BN. Evaluation of postoperative complications following elective surgeries of dogs and cats at private practices using computer records. Can Vet J 1996;37:672-678.
6. Kustritz MV. Determining the optimal age for gonadectomy of dogs and cats. J Am Vet Med Assoc 2007;231:1665-1675.
7. Adin CA. Complications of ovariohysterectomy and orchiectomy in companion animals. Vet Clin North Am Small Anim Pract 2011;41:1023-1039.
8. Howe LM. Surgical methods of contraception and sterilization. Theriogenology 2006;66:500-509.
9. Parkinson TJ. Progress towards less invasive veterinary surgery. Vet Rec 2012;171:67-68.