An update on anaplasmosis in dogs
Canine anaplasmosis is caused by one of two gram-negative, obligate, intracellular bacterial agents, Anaplasma phagocytophilum or Anaplasma platys. Both types are likely spread by ticks and can occur worldwide.
Canine anaplasmosis is caused by one of two gram-negative, obligate, intracellular bacterial agents, Anaplasma phagocytophilum or Anaplasma platys. Infection with A. phagocytophilum, the species more commonly associated with anaplasmosis, causes lameness and is often confused with Lyme disease. Infection with A. platys results in a cyclic thrombocytopenia. Both types of anaplasmosis are likely spread by ticks and can occur worldwide.

A NOTE ON CLASSIFICATION
Both A. phagocytophilum and A. platys are in the order Rickettsiales, which includes members of the genera Ehrlichia, Anaplasma, Cowdria, Wolbachia, and Neorickettsia. In 2001, a major restructuring of the classification of organisms occurred in the order Rickettsiales.1 As a result of these investigations, three separate species of Ehrlichia—Ehrlichia equi, Ehrlichia phagocytophila, and the previously unnamed agent causing human granulocytic ehrlichiosis—were found to be insufficiently different to warrant separate species designations. These organisms were phylogenetically most closely related to species in the genus Anaplasma, so they were reclassified as A. phagocytophilum. In addition, Ehrlichia platys was found to be more closely related to Anaplasma species and was subsequently renamed A. platys.1
ANAPLASMA PHAGOCYTOPHILUM INFECTION
A hallmark of this form of anaplasmosis is that it may result in subclinical disease or a chronic carrier state.
Epidemiology
Infection with A. phagocytophilum was first reported in dogs from Minnesota and Wisconsin in 1996.2 Anaplasma phagocytophilum infection is a vector-borne zoonotic disease, and its appearance in dogs in those areas closely coincided with recognition of the disease in people.3
The organism has a worldwide geographic distribution and is endemic in the upper Midwest, East, and Northeast regions of the United States as well as the western coastal regions. European countries such as the United Kingdom, Norway, Sweden, Switzerland, and Germany also have reported infections in ruminants, dogs, and people. The disease has been less frequently reported in Asia and South America. In the United States, most disease outbreaks are seasonal and coincide with the emergence of tick vectors in spring and early summer (May and June) and then again in the fall (September).
Tick vector and mammalian hosts
Several ixodid ticks can serve as vectors for A. phagocytophilum. In the United States, Ixodes scapularis is the primary vector in the upper Midwest and the Northeast, and Ixodes pacificus is the primary vector in the West. Ixodes ricinus is the primary vector in Europe. The organism can infect a wide range of mammals, including dogs, cats, horses, ruminants, people, and many wildlife species. White-tailed deer and several species of small rodents are considered the primary reservoir hosts. It is suspected that transmission to susceptible mammalian hosts requires prolonged tick attachment and feeding of 24 hours or more.4
Clinical and laboratory findings
Clinical disease in dogs is most often associated with the acute, bacteremic phase of infection. The duration and severity of the clinical findings during the acute phase of the disease can vary greatly and can last from one to several days.5
Chronic, subclinical, persistent infections have been documented for more than five-and-a-half months in dogs experimentally inoculated with a Swedish isolate of A. phagocytophilum and for almost one year in dogs inoculated with a human isolate from New York (NY18).6,7 However, no confirmed reports of clinical disease occurring in animals known to be chronic carriers exist. It is unknown whether a chronic carrier state can later result in chronic disease.
Clinical disease has most often been reported in dogs 8 years of age or older.2,8 Golden retrievers and Labrador retrievers are overrepresented in most reports, but it is uncertain whether this is the result of breed susceptibility, frequency of exposure in these breeds, or breed popularity.2,8,9
Clinical signs. A high seroprevalence in people and dogs living in endemic areas10 suggests that many infections result in a mild, flu-like disease that is self-limiting or in a subclinical infection. Animals with clinical disease associated with acute infection often have vague signs of illness including fever, lethargy, malaise, anorexia, and general muscle pain resulting in reluctance to move.
The most frequently observed clinical findings in dogs that would alert you to A. phagocytophilum infection are joint pain and lameness resulting from polyarthritis. Other less commonly observed clinical signs include gastrointestinal problems such as vomiting, diarrhea, or both or respiratory signs such as coughing and labored breathing. Central nervous system disease (meningitis) can also occur, resulting in seizure activity, ataxia, or neurologic manifestations such as dullness or stupor, but these findings are infrequently observed.
Because many animals present with clinical signs of polyarthritis and possibly a history of tick exposure, clinical signs of canine anaplasmosis may be indistinguishable from those seen with Lyme disease. In addition, both diseases are transmitted by the same tick vectors and have similar geographic distributions.
Laboratory test results. Abnormalities in laboratory test results may vary during the acute phase of the disease. Test results will likely be normal in persistently infected, subclinical carriers.
The most common hematologic abnormality noted in clinically affected dogs is a mild to severe thrombocytopenia, seen in more than 80% of acutely infected dogs.2,8,11 Additionally, many animals will initially have lymphopenia but later may develop a reactive lymphocytosis. Rarely, neutropenia is seen. Membrane-bound vacuoles of intracytoplasmic organisms (morulae) can sometimes be observed in circulating neutrophils and, rarely, in eosinophils in acutely infected dogs. A mild to moderate nonregenerative anemia is occasionally seen.
Common serum biochemical abnormalities include elevated serum alkaline phosphatase activity and mild to moderate hypoalbuminemia and hyperfibrinogenemia.2 The hypoalbuminemia usually resolves quickly once animals are afebrile.
Diagnosis
This form of anaplasmosis can be difficult to distinguish from Lyme disease, but new diagnostic capabilities are now available.

1. An intracytoplasmic Anaplasma phagocytophilum morula in a toxic band neutrophil in the peripheral blood of an infected dog (Wright's-Giemsa; 100X).
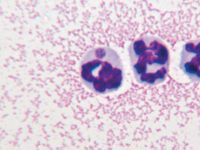
2. An intracytoplasmic Anaplasma phagocytophilum morula in a neutrophil in the synovial fluid of an infected dog (Wright's-GiemsaÃÂ; 100X).
Light microscopy. Canine anaplasmosis is often diagnosed by microscopic identification of morulae in circulating neutrophils in the peripheral blood (Figure 1) and sometimes the synovial fluid (Figure 2). These findings are most often identified during the acute phase of the disease. Experimentally, organisms appear in the peripheral blood between four and 14 days after inoculation and usually persist for up to eight days.5 Anaplasma phagocytophilum morulae may be seen in 1% to 27% of circulating neutrophils. The ability to identify morulae in the circulating neutrophils of infected dogs is enhanced by preparing and microscopically evaluating a buffy coat smear (Figure 3). In animals presenting with polyarthritis, synovial fluid analysis would reveal decreased viscosity and an increased leukocyte count (> 3,000 cells/µl) with a predominant neutrophil population. Organisms may also be found in a small number (≤ 1%) of neutrophils in synovial fluid (Figure 2).
Anaplasma phagocytophilum morulae cannot be distinguished from those caused by E. ewingii, the causative agent of canine granulocytic ehrlichiosis.12 Clinical signs of E. ewingii infection are also identical to those seen with canine anaplasmosis, and these two infections cannot be distinguished by clinical (fever, lethargy, lameness, or reluctance to stand) or routine laboratory (complete blood count or synovial fluid analysis) methods.
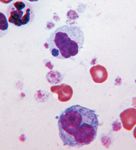
3. An intracytoplasmic Anaplasma phagocytophilum morula in a neutrophil (above center) in a buffy coat smear prepared from the peripheral blood of a dog. An intracytoplasmic Döhle body is seen in the neutrophil in the upper left corner (Wright's-Giemsa; 100X).
Serologic testing. Anaplasma phagocytophilum infection can be diagnosed serologically at most commercial laboratories with indirect fluorescent antibody (IFA) testing as well as with the in-house ELISA, SNAP 4Dx (IDEXX Laboratories).
The IFA test uses whole organisms grown in cell culture as the source of antigens. With this test, experimental studies have shown that seroconversion in dogs may occur as soon as two to five days after morulae appear in the peripheral blood.8 In another study using sera collected from confirmed cases of A. phagocytophilum infection in horses, dogs, people, and cattle, all serum samples were positive at titers of 1:80 or greater, and most had titers of ≥ 1:320.13 For this reason, and because nonspecific binding of antibodies can occur using this assay when serum samples are at dilutions of 1:40 or less, a titer of ≥ 1:80 is considered positive for antibodies to A. phagocytophilum. In cross-reactivity studies, serum samples from animals or people infected with Neorickettsia risticii, Ehrlichia canis, E. ewingii, Ehrlichia chaffeensis, Ehrlichia sennetsu, Rickettsia rickettsii, Ehrlichia typhi, Bartonella henselae, or Bartonella quintana did not contain antibodies that cross-reacted with A. phagocytophilum.13 Serum samples from animals infected with A. platys were not evaluated in this study.
The A. phagocytophilum test spot on the in-house SNAP 4Dx assay uses a synthetic peptide based on the immunodominant p44 protein of A. phagocytophilum as the antigen. Experimentally inoculated dogs had positive test results on the SNAP 4Dx assay as early as eight days after inoculation.7 These animals remained persistently infected for nearly one year and remained seropositive during that entire time even though the animals were clinically healthy and light microscopy did not reveal organisms in circulating neutrophils. In performance trials, the sensitivity and specificity of the SNAP 4Dx test for antibody to A. phagocytophilum was 99.4% and 100%, respectively.14 Dogs infected with Ehrlichia species, including E. ewingii, will not likely cross-react with the A. phagocytophilum test on the SNAP 4Dx. In addition to testing for infection with A. phagocytophilum, the SNAP 4Dx also detects infection with Dirofilaria immitis, Borrelia burgdorferi, or E. canis.
PCR testing. Nucleic acid amplification is the most sensitive method for detecting organism DNA in the peripheral blood of an infected animal. Several commercial laboratories offer polymerase chain reaction (PCR) analysis of peripheral blood for detecting A. phagocytophilum and E. ewingii. These assays are species-specific and can distinguish between these two infectious agents. Submit EDTA-anticoagulated peripheral blood to laboratories for PCR analysis. Collect these samples before initiating antimicrobial therapy because recent antibiotic administration, particularly tetracycline or its derivatives, increases the likelihood of a false negative result.
Although this technique is more sensitive in detecting circulating organisms than is light microscopy, it has been shown to be only intermittently positive in subclinically infected, persistent carriers of A. phagocytophilum since organisms may circulate intermittently in the peripheral blood of subclinically infected animals.7,15 Therefore, PCR analysis should not be considered a definitive method for conclusively excluding subclinical infection in clinically normal animals that are seropositive on routine screening.
Treatment and prognosis
The treatment for canine anaplasmosis is the same as that for infection with the closely related Ehrlichia species—doxycycline. The optimal dose and length of therapy have not been firmly established, but an oral dosage of 5 to 10 mg/kg given twice a day for 30 days has been recommended.12,16 In most cases, clinical signs resolve rapidly. Dogs are often markedly improved 24 to 48 hours after therapy is instituted, and the prognosis for clinical recovery is excellent.12 However, persistent infection and subclinical carrier states have been demonstrated in experimentally inoculated dogs treated with doxycycline for 14 days and 28 days at the recommended dose.7,15
Although clinical resolution of disease is usually accomplished, it may be difficult to ascertain the effectiveness of therapy in completely clearing organisms from naturally infected animals. If an animal no longer produces antibodies to the organism, this may indicate that the organism has been cleared from the body. Since PCR analysis can give false negative results, this assay is unreliable in detecting all subclinical, persistent carriers.
In light of these diagnostic challenges, once an animal with clinical anaplasmosis has been treated with the recommended course of doxycycline therapy and is clinically normal, administering a second course of doxycycline therapy or a different antimicrobial agent, in an attempt to obtain a seronegative status, is not recommended at this time.
Because tetracyclines may result in tooth discoloration, chloramphenicol (15 to 25 mg/kg every eight hours for 14 to 21 days) has been suggested as an alternative to tetracycline derivatives in dogs under 1 year of age.12 However, it appears that doxycycline, unlike tetracycline, does not result in enamel discoloration in young growing puppies.
In vitro testing has shown that rifampin and levofloxacin are also effective against A. phagocytophilum, but their effectiveness in vivo has not been evaluated in dogs.17
Seropositive, clinically healthy dogs
Animals from endemic areas are often exposed to A. phagocytophilum, and 40% or more of dogs in these areas may be seropositive.9 However, since the morbidity is low, it appears that many animals may have antibodies to A. phagocytophilum without having any concurrent evidence of clinical disease. Since persistent infection in clinically healthy dogs has been demonstrated,6,7,15 it is likely that a portion of the seropositive animals are chronically infected carriers of the organism. Experimentally, chronically infected carriers did not have any hematologic abnormalities,7 and, thus far, it appears that seropositive animals with no clinical evidence of disease are hematologically normal. Incidents of chronically infected carriers later developing clinical disease have not been clearly documented.
The cyclic appearance of clinical cases that coincide with tick season indicates that canine anaplasmosis is an acute disease that occurs in dogs a week or two after organism inoculation by ticks.5,12 Because chronic infection has not been directly related to clinical disease and because a therapeutic regimen effective in clearing the organism from an infected animal has not been established, treating clinically healthy, seropositive animals is of questionable benefit. However, a seropositive reaction to A. phagocytophilum in a clinically healthy dog should not be disregarded. At a minimum, implement an aggressive tick-control program designed to minimize exposure to ticks, and, hence, to other tick-borne diseases. It is clear that coinfection with two or more tick-borne agents is common and that dogs coinfected with B. burgdorferi and A. phagocytophilum are nearly two times more likely to develop clinical disease than are dogs infected with either agent alone.9 There is also some concern that chronically infected carriers could be adversely affected by therapeutic agents that compromise the immune system or by a concurrent illness that might alter an animal's immune status. The administration of immunosuppressive doses of corticosteroids to infected, asymptomatic dogs resulted in the reappearance of bacteremia, although the animals remained clinically normal.6,7,15
ANAPLASMA PLATYS INFECTION
Anaplasma platys (formerly Ehrlichia platys) is the causative agent of infectious cyclic thrombocytopenia in dogs.
Epidemiology
Anaplasma platys was first reported in the United States in 1978 and has since been recognized to have a worldwide distribution, being reported in many European, Asian, and South American countries.18 This agent is unique—it is the only intracellular infectious agent described in people or animals to specifically infect platelets (Figure 4).
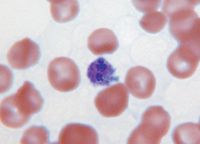
4. Two large, dark-blue-staining Anaplasma platys morulae in a circulating platelet from an infected dog. The normal, smaller, pink-staining platelet granules are also observed in the cytoplasm of the infected platelet (Wright's-GiemsaÃÂ; 100X).
Tick vector and mammalian hosts
The natural mode of disease transmission has not been conclusively determined, but A. platys DNA has been amplified from Rhipicephalus and Dermacentor species ticks.19,20 Therefore, tick transmission is highly suspected. Dogs are by far the most common mammalian host, although rare reports of infections in cats, impalas, and sheep have been documented outside the United States.21,22
Clinical and laboratory findings
Most dogs with naturally occurring infection have mild clinical disease, although more severe clinical signs of fever, lethargy, pale mucous membranes, petechial hemorrhages, epistaxis, and lymphadenopathy can occur. As with other tick-transmitted diseases, coinfection of A. platys with other infectious agents such as Ehrlichia, Bartonella, or Borrelia species can result in more severe clinical manifestations.
Experimentally, clinical signs appeared between eight and 14 days after inoculation.18 The organism appears in circulating platelets, resulting in a severe thrombocytopenia, typically < 20,000/µl. Platelet counts usually remain below 20,000/µl for only one or two days. The organisms disappear rapidly from the peripheral blood, and platelet counts rise quickly, reaching a normal value in three or four days. Subsequently, organisms will reappear at one- to two-week intervals, and the process repeats, resulting in a cyclic thrombocytopenia. The percentage of parasitized platelets and the severity of the thrombocytopenic episodes decrease with subsequent cycles of parasitemia.
Diagnosis
Anaplasma platys infection can be diagnosed by light microscopic identification of morulae in circulating platelets. An IFA test is also commercially available, and serologic testing has indicated that seropositivity in dogs can be high in the Southeast and Midwest.23 There appears to be some cross-reactivity between A. platys and the A. phagocytophilum with the SNAP 4Dx assay. With this assay, many animals infected with A. platys will have positive test results for A. phagocytophilum. The organisms are closely related and evidently share epitopes used in this ELISA. PCR testing for A. platys, which can distinguish between the two organisms, is typically done in experimental laboratories for research purposes, but a PCR test is also commercially available (e.g. Zoologix).
Treatment and prognosis
Doxycycline at the dosage described above for A. phagocytophilum is apparently effective in treating A. platys infection.12 However, animals with circulating A. platys organisms that have moderate to severe clinical disease or that do not rapidly respond to doxycycline therapy should be tested for other tick-borne diseases.
SUMMARY
Anaplasmosis causes one of two clinical syndromes: Fever, lethargy, polyarthritis, and, less often, gastrointestinal, respiratory, or neurologic signs, which are associated with A. phagocytophilum infection. Signs due to thrombocytopenia are associated with A. platys infection.
Since the two organisms share the same tick vector, the diseases have a similar geographic distribution. Anaplasma phagocytophilum infection is an emerging tick-borne infection in dogs, endemic to several areas of the United States and expanding in distribution. The organism typically causes acute clinical disease similar to Lyme disease, resulting in a suppurative polyarthritis. In addition to causing clinical disease, A. phagocytophilum may persistently infect dogs, causing a subclinical carrier state associated with positive serologic test results in a healthy dog. These dogs should be monitored for evidence of clinical disease because there may be potential for future clinical manifestations, particularly if they become coinfected with other tick-borne agents or become immunosuppressed. Since animals can have subclinical infections, it is important to recognize that clinically ill animals may have positive test results for A. phagocytophilum but be experiencing the clinical manifestations of an unrelated disease process. Therefore, seropositivity alone does not indicate a causal relationship to the clinical disease. Clinical signs must be consistent with those found in animals with anaplasmosis and should rapidly resolve in most cases shortly after doxycycline therapy is initiated.
Dogs infected with A. platys experience a cyclic thrombocytopenia. Clinical disease is often mild in naturally infected dogs, but some animals may have clinical evidence of bleeding (epistaxis or petechiation), particularly during the initial bacteremic phase when platelet counts may be reduced to < 20,000 cells/µl. Platelet counts are less dramatically affected in subsequent cycles of bacteremia and clinical signs become milder or unapparent. As with A. phagocytophilum infection, animals with clinical disease resulting from A. platys infection respond rapidly to treatment with doxycycline.
Finally, A. phagocytophilum is a zoonotic pathogen. However, direct transmission from animals to people or animal to animal is highly unlikely and, to our knowledge, has never been documented. Even so, alert owners of pets with known exposure to A. phagocytophilum to potential human exposure from infected ticks in the environment.
Editors' note: IDEXX Laboratories has sponsored presentations by both Dr. Alleman and Dr. Wamsley at various continuing education symposia.
A. Rick Alleman, DVM, PhD, DABVP, DACVP
Heather L. Wamsley, DVM, DACVP
Department of Physiological Sciences
College of Veterinary Medicine
University of Florida
Gainesville, FL 32608
REFERENCES
1. Dumler JS, Barbet AF, Bekker CP, et al. Reorganization of genera in the families Rickettsiaceae and Anaplasmataceae in the order Rickettsiales: unification of some species of Ehrlichia with Anaplasma, Cowdria with Ehrlichia and Ehrlichia with Neorickettsia, descriptions of six new species combinations and designation of Ehrlichia equi and 'HGE agent' as subjective synonyms of Ehrlichia phagocytophila. Int J Sys Evol Microbiol 2001;51(Pt 6):2145–2165.
2. Greig B, Asanovich KM, Armstrong PJ, et al. Geographic, clinical, serologic, and molecular evidence of granulocytic ehrlichiosis, a likely zoonotic disease, in Minnesota and Wisconsin dogs. J Clin Microbiol 1996;34(1):44-48.
3. Bakken JS, Dumler JS, Chen SM, et al. Human granulocytic ehrlichiosis in the upper Midwest United States. A new species emerging? JAMA 1994;272(3):212-218.
4. des Vignes F, Piesman J, Heffernan R, et al. Effect of tick removal on transmission of Borrelia burgdorferi and Ehrlichia phagocytophila by Ixodes scapularis nymphs. J Infect Dis 2001;183(5):773-778.
5. Egenvall A, Bjöersdorff A, Lilliehöök I, et al. Early manifestations of granulocytic ehrlichiosis in dogs inoculated experimentally with a Swedish Ehrlichia species isolate. Vet Rec 1998;143(15):412-417.
6. Egenvall A, Lilliehöök I, Bjöersdorff A, et al. Detection of granulocytic Ehrlichia species DNA by PCR in persistently infected dogs. Vet Rec 2000;146(7):186-190.
7. Alleman AR, Chandrashekar R, Beall M, et al. Experimental inoculation of dogs with a human isolate (Ny18) of Anaplasma phagocytophilum and demonstration of persistent infection following doxycycline therapy (abst). J Vet Intern Med 2006;20:763.
8. Egenvall AE, Hedhammar AA, Bjöersdorff AI. Clinical features and serology of 14 dogs affected by granulocytic ehrlichiosis in Sweden. Vet Rec 1997;140(9):222-226.
9. Beall MJ, Chandrashekar R, Eberts MD, et al. Serological and molecular prevalence of Borrelia burgdorferi, Anaplasma phagocytophilum, and Ehrlichia species in dogs from Minnesota. Vector Borne Zoonotic Dis 2008 Feb 27 [Epub ahead of print].
10. Bakken JS, Goellner P, Van Etten M, et al. Seroprevalence of human granulocytic ehrlichiosis among permanent residents of northwestern Wisconsin. Clin Infect Dis 1998;27(6):1491-1496.
11. Lilliehöök I, Egenvall A, Twedten HW. Hematopathology in dogs experimentally infected with a Swedish granulocytic Ehrlichia species. Vet Clin Pathol 1998;27(4):116-122.
12. Greig B, Armstrong PJ. Canine granulocytotropic anaplasmosis (A. phagocytophilum infection). In: Greene CE, ed. Infectious diseases of the dog and cat. 3rd ed. St. Louis, Mo: Saunders, 2006;219-224.
13. Dumler JS, Asanovich KM, Bakken JS, et al. Serologic cross-reactions among Ehrlichia equi, Ehrlichia phagocytophila, and human granulocytic Ehrlichia. J Clin Microbiol 1995;33(5):1098-1103.
14. Chandrashekar R, Mainville C, Daniluk D, et al. Performance of an in-clinic test SNAP 4Dx, for the detection of antibodies to canine granulocytic infection, Anaplasma phagocytophilum (abst). J Vet Intern Med 2007;21:626.
15. Alleman AR, Wamsley HL, Abbott J, et al. Experimental Anaplasma phagocytophilum infection of dogs by intravenous inoculation of human and canine isolates and treatment with doxycycline (abst). Vet Pathol 2007;44:19.
16. Breitschwerdt EB, Hegarty BC, Hancock SI. Sequential evaluation of dogs naturally infected with Ehrlichia canis, Ehrlichia chaffeensis, Ehrlichia equi, Ehrlichia ewingii, or Bartonella vinsonii. J Clin Microbiol 1998;36(9):2645-2651.
17. Maurin M, Bakken JS, Dumler JS. Antibiotic susceptibilities of Anaplasma (Ehrlichia) phagocytophilum strains from various geographic areas in the United States. Antimicrob Agents Chemother 2003;47(1):413-415.
18. Harvey JW, Simpson CF, Gaskin JM. Cyclic thrombocytopenia induced by a Rickettsia-like agent in dogs. J Infect Dis 1978;137(2):182-188.
19. Inokuma H, Raoult D, Brouqui P. Detection of Ehrlichia platys DNA in brown dog ticks (Rhipicephalus sanguineous) in Okinawa Island, Japan. J Clin Microbiol 2000;38(11):4219-4221.
20. Parola P, Cornet JP, Sanogo YO, et al. Detection of Ehrlichia spp., Anaplasma spp., Rickettsia spp., and other eubacteria in ticks from the Thai-Myanmar border and Vietnam. J Clin Microbiol 2003;41(4):1600-1608.
21. Santarém VA, Laposy CB, Farias MR. Ehrlichia platys-like inclusions and morulae in platelets of a cat (abst). Brazilian J Vet Sci 2000;7:130.
22. Du Plessis L, Reyers F, Stevens K. Morphological evidence for infection of impala, Aepyceros melampus, platelets by a rickettsia-like organism. Onderstepoort J Vet Res 1997;64(4):317-318.
23. Hoskins JD, Breitschwerdt EB, Gaunt SD, et al. Antibodies to Ehrlichia canis, Ehrlichia platys, and spotted fever group rickettsiae in Louisiana dogs. J Vet Intern Med 1988;2(2):55-59.