What's your diagnosis? Purulent upper respiratory discharge in a rabbit
After an enucleation, purulent material began coming from this rabbit's nose and filled the empty orbit. Find out why.
A 2.5-year-old castrated male mixed-breed rabbit was referred to North Carolina State University's Veterinary Hospital for evaluation of purulent discharge from the nares and from the site of a recent enucleation.
HISTORY
Starting at 18 months of age, the rabbit had been treated by the referring veterinarians with ocular corticosteroids and antibiotics for left-sided ocular discharge, which progressed to include periocular swelling and corneal ulcers. The eye had been enucleated one month before presentation. Soon after surgery, the orbit filled with purulent material, and the rabbit developed purulent nasal discharge.
The rabbit's appetite was reportedly good until after the enucleation, when it began to gradually decline. The owners had been attempting to syringe feed (Critical Care-Oxbow Animal Health) in the days preceding their appointment but had been having difficulty.
PHYSICAL EXAMINATION
At presentation, the rabbit was orthopneic and open-mouth breathing; both nares were occluded with purulent discharge. There was significant periorbital swelling on the left side, and purulent material could be expressed from the incompletely healed enucleation site.
Quick tip
After a brief evaluation, consider lightly sedating dyspneic small mammals before performing a full examination. A wide variety of sedation protocols are reported in the literature,5 but we find that a combination of butorphanol (0.2 to 0.4 mg/kg) and midazolam (0.5 to 1 mg/kg) intramuscularly or subcutaneously often works well for these patients.
A brief oral examination performed with an otoscope revealed halitosis, a discolored right maxillary incisor and mild elongation of the left mandibular molars with mild lingual points. The rabbit was in poor body condition, had decreased gastrointestinal sounds on auscultation and produced scant feces during hospitalization.
On physical examination, the rabbit had purulent rhinitis and dacryocystitis, disease processes that are commonly associated with underlying dental disease in rabbits.1-4 Although only mild abnormalities were noted during this rabbit's oral examination, underlying dental disease was still a top differential diagnosis. This rabbit also had evidence of gastrointestinal stasis, presumably secondary to stress and decreased food intake.
DIAGNOSTIC TESTS
Preanesthetic blood work revealed anemia (23%; reference range = 30% to 50%) and a normal white blood cell count (5 x 103/µl; reference range = 5 to 12 x 103/µl) with relative heterophilia (60%; reference range = 35% to 55%) and lymphopenia (20%; reference range = 25% to 60%). All serum chemistry profile values were within published normal ranges.5
On whole body radiographs (Figure 1), the roots of multiple maxillary cheek teeth were poorly defined, with expansile lesions consistent with abscessation (white arrows). The occlusal surfaces of the cheek teeth were uneven (a finding that was not appreciated on the conscious oral examination). There was subtle increased opacity of the left tympanic cavity and thickening of the tympanic bulla suggestive of otitis media.
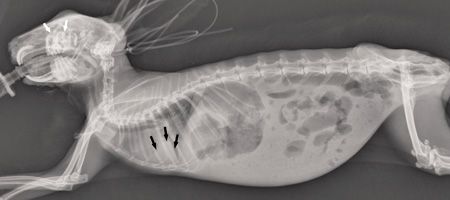
1. A whole body radiograph of the rabbit in this case. The roots of multiple maxillary cheek teeth were poorly defined, with expansile lesions consistent with abscessation (white arrows), and the occlusal surfaces of the cheek teeth were uneven. In the thorax, there was a diffuse interstitial pattern and a patchy, ventrally distributed alveolar pattern, consistent with aspiration pneumonia (black arrows).
In the thorax, there was a diffuse interstitial pattern and a patchy, ventrally distributed alveolar pattern, consistent with aspiration pneumonia (black arrows). The stomach was gas-dilated, consistent with aerophagia secondary to dyspnea. Gastrointestinal fill was subjectively decreased, and large pockets of gas were present in the cecum and intestines, consistent with gastrointestinal stasis syndrome. Serosal detail was decreased, most likely because of decreased abdominal fat stores.
Computed tomography (CT) confirmed extensive destruction of the roots of all the maxillary cheek teeth with osteomyelitis of the surrounding bone (Figure 2). The maxillary bone had expanded into the pterygopalatine fossa on the left side, causing a retrobulbar mass effect (thick white arrow). This effect likely contributed to the ocular disease that prompted the referring veterinarian to perform the enucleation.
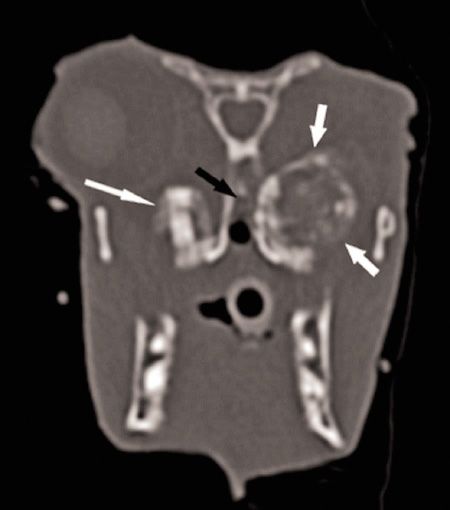
2. Transverse CT image of the skull at the level of the eyes. There is a large, expansile lesion affecting the left maxillary molar and extending into the retrobulbar space (thick white arrows). The left eye is absent. Note that the right maxillary molar is also affected (thin white arrow) and the nasal cavity is partially occluded (black arrow).
The CT also confirmed the clinical suspicion that the nasal passages were completely obstructed and the nasal turbinates had been destroyed by severe rhinitis. Fluid was noted in the left bulla, further supporting a diagnosis of left-sided otitis media.
OUTCOME
Because of the degree of dyspnea and the grave prognosis associated with such severe dental disease, the rabbit was humanely euthanized. Necropsy confirmed abscessation of the roots of all the maxillary cheek teeth. The nasal passages were occluded with purulent material, and the architecture of the nasal turbinates was destroyed (Figure 3). Marked otitis media and interna were noted in the left ear. The rabbit also had severe aspiration pneumonia, with more than 50% of the lung volume affected.
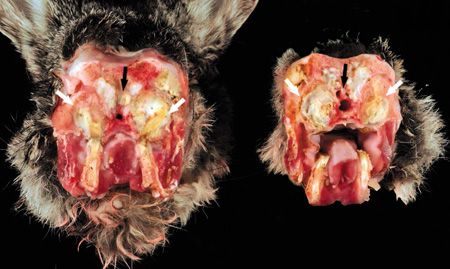
3. Photos of the skull taken at necropsy with the skull sectioned just caudal to the eyes. Multifocal abscessation of the maxillary cheek teeth is apparent (white arrows). The nasal passages are partially occluded with purulent material (black arrows).
Typically, a full anesthetized oral examination would have been performed using an oral speculum and cheek dilators to improve visualization of the cheek teeth. In this case, because the owners chose to euthanize, this examination was performed postmortem and revealed minor unevenness of the occlusal surfaces, but no other significant findings.
DISCUSSION
Dental disease is common in rabbits and can encompass many disease processes, including malocclusion, elongation and tooth root abscessation.1,3 Dental disease can be associated with a number of secondary problems, including dacryocystitis, exophthalmos, rhinitis and sinusitis.1-4
Key points
1. Ocular and upper respiratory disease in rabbits is often associated with underlying dental disease.
2. A normal oral examination does not exclude dental disease as a possible cause of a patient's clinical signs.
3. Use of sedation is strongly recommended for the examination and imaging of small mammals that are in respiratory distress. If these patients are anesthetized, their airway should be protected.
4. While radiography allows for the preliminary assessment of dental health in rabbits, CT is strongly recommended for full evaluation of dental, upper respiratory and ocular disorders.
5. In severe cases of dental-associated respiratory, ocular or otic disease, medical treatment alone is rarely successful. Surgical intervention is almost always required to improve a patient's clinical status. Even with aggressive surgical treatment, the prognosis is fair to grave, depending on the severity of disease.
6. Any disease process that causes pain or stress in rabbits can be complicated by secondary gastrointestinal stasis. Providing nutritional support to a dyspneic rabbit can be challenging and carries a high risk of aspiration.
Given the progression of clinical signs and resulting pathology in this case, it is suspected that the rabbit's primary disease process was tooth root abscessation, with ocular and respiratory signs developing secondarily. One of the abscesses had created a left retrobulbar mass effect, which most likely caused exophthalmos and secondary corneal ulceration. The ocular discharge could have been secondary to impingement on the nasolacrimal duct by the abscess or an extension of the disease through the duct. As the severity of the dental disease progressed, purulent material likely extended dorsally into the nasal cavity and possibly through the auditory tube into the left tympanic bulla. Enucleation may have contributed to the upper respiratory obstruction because closing the orbit eliminated a route for drainage of purulent material through the nasolacrimal duct.
Because rabbits are obligate nasal breathers,6 rhinitis compromises a rabbit's respiratory status more than it compromises a dog's or a cat's. The pulmonary disease can be attributed to inhalation of purulent material from the nasal passages or aspiration of food material from assisted feeding.
Despite severe abscessation of all maxillary cheek teeth, this rabbit had a relatively unremarkable oral examination (even on necropsy) and had not exhibited changes in appetite until late in the disease's progression. This case illustrates the importance of performing imaging in all rabbits with upper respiratory clinical signs and most rabbits with ocular clinical signs, even if more classic clinical signs of dental disease (anorexia, ptyalism) are not present. In this rabbit, whole body radiography was also recommended to assess the lungs and the gastrointestinal tract.
While dental abnormalities were apparent on radiographs, CT allowed for a much more detailed evaluation of the extent of disease.7 CT is the preferred imaging modality for full evaluation of dental and upper respiratory anatomy and should be considered for all rabbits with suspected tooth root disease.1,3,4
For practitioners who do not have access to this technology or clients who are unable to afford it, a six-view skull radiograph series taken with mammography or dental film can provide more detailed information than the two-view whole body radiographs shown here. However, these images can be challenging to acquire and interpret for practitioners who do not have extensive experience with them, and CT images are often more intuitive to interpret. Radiographs also provide much less detailed information about the upper airway and the retrobulbar space, which were important in this case.8
Treatment of odontogenic abscesses and their sequelae is different in each case, but these patients generally require surgical intervention, including abscess débridement, tooth extraction or rhinotomy.1,3,4 Because rabbit heterophils contain lower activities of myeloperoxidase than other mammals', their purulent exudate is thicker and more tenacious.9
Medical therapy alone is rarely successful, but antibiotic therapy may be helpful as an adjunct to surgery. It is recommended that antibiotics be selected based on culture and sensitivity performed on samples taken at the time of surgery. A recent study suggests that penicillin (given parenterally only, never orally) or chloramphenicol may be good empirical choices, pending culture results.10 Even with aggressive management, owners should be aware that procedures are palliative and complete cure is rare.
Nutritional support is also critical for these patients to prevent secondary gastrointestinal stasis. Unfortunately, syringe feeding is often stressful for the dyspneic patient and carries a high risk of aspiration pneumonia. Placement of a nasogastric tube can be considered but may contribute to respiratory distress in a patient whose upper airway is already partially obstructed.11
Samantha D. Swisher, DVM
Avian & Exotic Animal Clinic of Indianapolis
9330 Waldemar Road
Indianapolis, IN 46268
Vanessa L. Grunkemeyer, DVM, DABVP (avian)
College of Life Sciences and Agriculture
University of New Hampshire
Durham, NH 03824
Mason Y. Savage, DVM
Department of Molecular Biomedical Sciences
College of Veterinary Medicine
North Carolina State University
Raleigh, NC 27607
Jessica R. Durrant, MS, DVM
Luke B. Borst, DVM, PhD, DACVP
Department of Population Health and Pathobiology
College of Veterinary Medicine
North Carolina State University
Raleigh, NC 27607
References
1. Capello V, Lennox A. Small mammal dentistry. In: Quesenberry KE, Carpenter JW, eds. Ferrets, rabbits, and rodents: clinical medicine and surgery. 3rd ed. St. Louis: Saunders, 2012;452-471.
2. Florin M, Rusanen E, Haessig M, et al. Clinical presentation, treatment, and outcome of dacryocystitis in rabbits: a retrospective study of 28 cases (2003-2007). Vet Ophthalmol 2009;6:350-356.
3. Reiter AM. Pathophysiology of dental disease in the rabbit, guinea pig, and chinchilla. J Exotic Pet Med 2008;17:70-77.
4. Lennox A. Rhinotomy and rhinostomy for surgical treatment of chronic rhinitis in two rabbits. J Exotic Pet Med 2013;22:383-392.
5. Carpenter JW, Marion CJ. Exotic animal formulary. 4th ed. St. Louis: Saunders, 2013;452.
6. Varga M. Cardiorespiratory disease. In: Textbook of rabbit medicine. 2nd ed. Oxford, England: Butterworth-Heinemann, 2013;390.
7. Van Caelenberg AI, De Rycke LM, Hermans K, et al. Comparison of radiography and CT to identify changes in the skulls of four rabbits with dental disease. J Vet Dent 2011;28(3):172-181.
8. Capello V, Gracis M, Lennox A. Rabbit and rodent dentistry handbook. Hoboken, New Jersey: Wiley Blackwell, 2005; 65-75.
9. Varga M. Abscesses. In: Textbook of rabbit medicine. 2nd ed. Oxford, England: Butterworth-Heinemann, 2013; 249.
10. Tyrrell KL, Citron DM, Jenkins JR, et al. Periodontal bacteria in rabbit mandibular and maxillary abscesses. J Clin Microbiol 2002;40(3):1044–1047.
11. Rosen L. Nasogastric tube placement in rabbits. J Exotic Pet Med 2011;20:27-31.
UN, WHO address public health concern over avian flu transmission to humans
April 18th 2024Veterinary professionals working with certain animals are advised to take precautionary steps to minimize risk of infection, while researchers in Texas study potential H5N1 vaccines, antivirals, and antibody therapies for humans
Read More