Comparing chemical restraint and anesthetic protocols used for blood donations in cats: One teaching hospital's experience
From 1998 to 2003, we evaluated several chemical restraint protocols for blood donor cats to improve the quality and efficiency of the blood bank, as well as the donors' quality of life.
The Ohio State University Veterinary Teaching Hospital has had an organized blood bank for small-animal transfusion medicine for more than 20 years. We currently process one to two units of feline blood a week and store both packed red blood cells and fresh frozen plasma.
Before 1998, cats were housed at the hospital to be on-demand donors of whole fresh blood. Because of the nature of on-demand donation, cats were not typically fasted at the time of blood collection, and the choice of chemical restraint (sedatives and anesthetics) was based primarily on time, expense, and ease of administration. During those years, intramuscular injections of ketamine hydrochloride were used with minimal complications. However, several aspects of chemical restraint with ketamine as a sole restraint agent were undesirable, such as the pain associated with administration, the extended recovery periods, and the worsening fractiousness of the cats with each subsequent donation. From 1998 to 2003, we evaluated several chemical restraint protocols for blood donor cats to improve the quality and efficiency of the blood bank, as well as the donors' quality of life.
We investigated the use of various chemical restraint protocols to facilitate quicker recovery times with minimal adverse effects, since the donors now have a scheduled time for donation, are appropriately fasted, and are to be sent home with the owners as soon as they recover. In this article, we review the advantages and disadvantages of the various methods of chemical restraint used by The Ohio State University Transfusion Medicine Service. We evaluated ketamine, midazolam hydrochloride and ketamine, medetomidine hydrochloride and atipamezole hydrochloride, acepromazine maleate and butorphanol tartrate, and sevoflurane. We chose not to evaluate propofol, another commonly used induction agent in private practice, because it has a short duration of action, it is best administered by intravenous catheter, it can induce apnea, and it has a high cost per average dose.
The subjective criteria used by the Transfusion Medicine Service staff to evaluate these restraint protocols are listed in Table 1. Table 2 presents additional information on the chemical restraint protocols we evaluated. A blood collection was considered successful if a minimum of 50 ml blood was collected.

TABLE 1: Criteria for Evaluating Chemical Restraint Protocols for Feline Blood Collection
BLOOD COLLECTION PROCEDURE
Blood donors were neutered males and spayed females between 2 and 7 years old. Twenty to 30 cats were enrolled in the donation program at any given time. The cats weighed at least 8 lb (3.6 kg) and had negative test results for feline leukemia virus, feline immunodeficiency virus, and heartworm infections. Most had type-A blood, though a limited number of type-B cats were also used in the program. Complete blood count and serum chemistry profile results and thyroid hormone concentrations for all donors were within reference ranges for our laboratory.
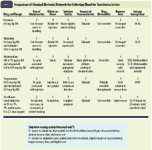
TABLE 2: Comparison of Chemical Restraint Protocols for Collecting Blood for Transfusion in Cats
For each collection, the cat was sedated or anesthetized, the hair over the jugular vein was clipped, and the site was scrubbed with chlorhexidine and cleaned with isopropyl alcohol. Each cat was placed on a circulating warm-water blanket, and ophthalmic lubricant was administered. A 20-ga butterfly catheter was placed in the jugular vein, and the blood was collected into a 60-ml syringe containing 8 ml acid citrate dextrose (ACD) solution. The blood was then transferred to a blood collection bag and separated into plasma and packed red blood cells. On rare occasions, the blood was used as whole fresh blood. Lactated Ringer's solution (50 ml) was administered subcutaneously to each donor immediately after donation. The cats' heart and respiratory rates and capillary refill times were monitored during donation and recovery.
KETAMINE
Ketamine (100 mg/ml) was given at a dose of 10 mg/kg intramuscularly. Ketamine is a nonbarbiturate anesthetic widely used alone or in combination with other drugs (particularly tranquilizers, barbiturates, or benzodiazepines).1 It produces a state in which the patient is dissociated from the environment, is immobile, and does not respond to pain; however, it is not a potent analgesic.2 Ketamine is an appropriate anesthetic and sedative agent for short diagnostic and minor surgical procedures in cats, especially those that do not require skeletal muscle relaxation.3 Characteristics of sedation with ketamine include increased muscle tone and an apneustic breathing pattern.1 Ketamine also preserves the swallowing and palpebral reflexes, and cats are typically hyperresponsive and ataxic during recovery.1 Ketamine is eliminated almost exclusively by the kidneys, with fecal elimination accounting for less than 5% of drug metabolism.4
Studies have shown that intramuscular ketamine mixed with xylazine hydrochloride causes muscle necrosis and is not recommended for use in survival procedures involving laboratory rodents.5,6 A similar situation may occur in cats.
Advantages
Ketamine was our chemical restraint method of choice for blood collection for years, as it is fairly inexpensive, accessible, and easy to administer. Ketamine increases heart rate and blood pressure and produces minimal vasoconstriction. These cardiovascular effects permit efficient blood collection.
Disadvantages
Our donors became counterconditioned to being handled for subsequent donations. They appeared to associate the handling with pain. Ketamine also carries a risk of causing muscular necrosis.
It took 20 to 45 minutes for ketamine to induce an appropriate level of sedation when given intramuscularly. It was unknown why it took so long to reach satisfactory sedation. The cats were adequately sedated during collection, but they were hyperesthetic when handled. Muscle jerks and repeated swallowing sometimes made needle positioning difficult and caused some blood collections to be prematurely aborted.
Recovery times were prolonged, usually lasting six to eight hours and, in one case, 24 hours. Occasional violent emergence behavior was noted in several cats (e.g. growling, biting, scratching, lunging at the cage). The cats were occasionally hypothermic immediately after donation (never less than 98 F [36.7 C]). Tachyphylaxis occurred in one donor after years of receiving the same ketamine dose before blood collection (the cat had received ketamine no more frequently than every three weeks).
MIDAZOLAM AND KETAMINE
Midazolam (5 mg/ml) was given at a dose of 0.4 mg/kg intramuscularly in the same syringe with ketamine at a dose of 4 to 6 mg/kg intramuscularly. Only one cat required a second midazolam dose (0.4 mg/kg) because of inadequate muscle relaxation. Midazolam is a short-acting benzodiazepine.1,7 It causes mild tranquilization by enhancing the effect of central nervous system (CNS) inhibitory neurotransmitters, opening chloride channels, and acting upon benzodiazepine receptors.1 In people, benzodiazepines act at the limbic, thalamic, and hypothalamic levels of the CNS, producing anxiolytic, sedative, hypnotic, skeletal muscle relaxant, and anticonvulsant effects.8
Midazolam is often used in veterinary medicine as a water-soluble alternative to diazepam, particularly to provide skeletal muscle relaxation in combination with ketamine. Midazolam is primarily metabolized by the liver into two hydroxylated metabolites, which are then excreted by the kidneys.9 Midazolam was selected instead of diazepam based on its efficacy when given intramuscularly as well as the anesthesiologists' recommendations at The Ohio State University Veterinary Teaching Hospital.
Advantages
Midazolam is readily absorbed from muscle. The cats were less reactive to the injection of midazolam and ketamine when compared with ketamine alone. The quality of restraint was exceptional, as the muscle jerks and hyperesthesia seen when using only ketamine were alleviated or eliminated. The cardiovascular effects of ketamine were still present, allowing for quick and efficient blood collections.
Disadvantages
It is known that the pharmacologic effects of midazolam can be seen clinically within five to 20 minutes,8 but in our experience maximal effects did not occur until 30 to 60 minutes after an intramuscular injection. The elimination half-life of midazolam alone in people is about two hours.10 Clinically, we noted that cats continued to be ataxic, unresponsive, and recumbent for as long as four to six hours after phlebotomy when we used the ketamine-midazolam combination. The combination also costs more than ketamine alone (Table 2).
MEDETOMIDINE AND ATIPAMEZOLE REVERSAL
Medetomidine (1 mg/ml) was given at a dose of 40 to 50 μg/kg intramuscularly. Medetomidine is an alpha2-adrenergic agonist that produces sedation by stimulating alpha2 receptors, thereby decreasing norepinephrine release.1 A sleeplike state that can be readily reversed with an alpha2 antagonist is produced. Atipamezole is an alpha2-adrenergic antagonist that directly inhibits the actions of medetomidine. Immediately after blood collection, subcutaneous fluids were given, and then atipamezole (5 mg/ml) was administered at a dose of 0.1 to 0.15 mg/kg intramuscularly.
Advantages
Cats were minimally reactive when the drug was injected intramuscularly. It took 10 to 20 minutes for medetomidine to produce adequate sedation. Drug reversal with atipamezole facilitated quick recoveries.
Disadvantages
Medetomidine was initially selected for evaluation in our donor program because it provides appropriate levels of sedation, is reversible, and allows quick recoveries. However, we found medetomidine unsatisfactory for use in cats during blood collection. Medetomidine results in marked bradycardia and increased peripheral vascular resistance, and it decreases cardiac output by 30% to 50%.1 This is undesirable during phlebotomy; several cats became pale and bradycardic (heart rates of 40 to 60 beats/min) with this protocol. Moreover, vasoconstriction made venipuncture and blood collection difficult. Several procedures had to be aborted because of inadequate blood flow into the collection system. In addition, medetomidine invariably caused emesis within five minutes of intramuscular administration. Medetomidine with or without atipamezole has a high cost per average dose (Table 2).
Atropine sulfate (0.4 mg/ml), an anticholinergic, was added to this protocol later as a presedative (0.02 mg/kg intramuscularly) to improve cardiac output. When using this combination, we noted that the quality of sedation was insufficient and unpredictable, although we failed to find an explanation for this phenomenon. Possible risks associated with using an anticholinergic with medetomidine include arrhythmias, hypertension, and increased myocardial workload. We found little benefit with adding atropine and do not recommend its use in cases such as this.
We added ketamine (4 to 6 mg/kg given intramuscularly in the same syringe as medetomidine) to this protocol11-16 in an attempt to improve cardiac output, but this resulted in prolonged recovery (a few hours in some cases), even with reversal of the alpha2 agonist, and most cats reacted painfully to the injection. The combination of ketamine and medetomidine potentially increases the risk of hypertension and increased systemic vascular resistance.
ACEPROMAZINE AND BUTORPHANOL
Acepromazine (10 mg/ml) was given at a dose of 0.05 mg/kg intramuscularly in the same syringe with butorphanol (10 mg/ml) at a dosage of 0.4 mg/kg intramuscularly. Acepromazine is a phenothiazine tranquilizer that acts by depressing the reticular activating system and suppressing the sympathetic nervous system.1 It also has a marked antiemetic effect by inhibiting dopamine in the chemoreceptor trigger zone.1 Butorphanol is an opioid that exhibits kappa agonist and mu antagonist effects, as it binds with opioid receptors in the CNS to produce analgesia and sedation.1 Opioids are commonly used with tranquilizers to produce neuroleptanalgesia (a state of tranquility, altered awareness, and analgesia) and marked sedation.
Advantages
Cats were minimally reactive when the drugs were injected intramuscularly. Reversal of the drug combination was never required.
Disadvantages
The combination did not appear to affect the senses of touch, sight, or hearing at the observed dosages. Therefore, it did not provide enough sedation for phlebotomy and was not cost-effective (Table 2). Blood collection was attempted but was unsuccessful (< 50 ml blood collected).
SEVOFLURANE
Sevoflurane was given by mask at 4% to 5% for induction and 4% for maintenance with oxygen at a rate of 1 to 2 L/min. Sevoflurane is an inhalant anesthetic that has been approved in the United States for use in people since 1995.17 It has chemical properties similar to those of isoflurane but has a higher minimum alveolar concentration (MAC; the potency of an inhalation anesthetic, based on the concentration necessary to produce no response in 50% of patients exposed to a painful stimulus).1 In practical terms, this means that sevoflurane is less soluble in blood and, therefore, allows more control over anesthetic depth and a more rapid recovery time.17,18
When compared with other inhalants, such as halothane or isoflurane in people, sevoflurane has minimal cardiovascular effects such as arrhythmias, myocardial depression, and hypotension.17 It is also less irritating to the airways.17 Researchers have shown that arterial blood pressures in cats during sevoflurane-oxygen anesthesia were significantly higher than in those receiving either isoflurane- or halothane-oxygen anesthesia.19 However, other studies have shown that hypotension can occur with sevoflurane at twice the MAC, or a surgical plane of anesthesia, as well as with the other common inhalants.19 Like all inhalant anesthetics, excretion of sevoflurane occurs in the lungs and depends on pulmonary ventilation, blood flow, and drug solubility.1
Advantages
Administration was painless and did not produce emesis or a prolonged excitatory phase. Induction was rapid, as an appropriate level of anesthesia was obtained in one to three minutes. Because cardiac output and peripheral perfusion are maintained, venipuncture and blood collection were smooth and efficient, with collection times ranging from five to 10 minutes. The cats were in a true state of anesthesia and were not responsive to environmental stimuli. This was safer for both the handler and the cat.
Recovery was rapid and was associated with minimal to no excitation. Cats were bright and alert within one hour. We also noted that cats were active and wanted to eat within an hour of recovering, and they did not vomit their food.
Disadvantages
Mask induction was stressful for a few of the more tense and aggressive cats. Preanesthetics were considered, but the benefits of rapid induction and recovery outweighed the disadvantage of occasional stress. In addition, gas inhalants are not reversible, and sevoflurane costs more than other gas inhalants (Table 2). Environmental exposure of the phlebotomist and handler was also a concern, even with an appropriate scavenging system. This protocol should not be used by pregnant women or women attempting to become pregnant. The sequelae of long-term exposure to low levels of sevoflurane are largely unknown. We were also concerned about inducing and maintaining anesthesia without securing an airway. An endotracheal tube was always available in case we needed intubation once we reached an appropriate depth of anesthesia with sevoflurane administered through the mask. However, intubation was never required.
CONCLUSION
After evaluating these different techniques, we considered inhalant anesthesia with sevoflurane to be the most reliable method for immobilizing the cats for blood donation. We also found that administering the inhalant anesthetic through a mask did not interfere with blood collection. Although not specifically studied in this paper, isoflurane would be an acceptable alternative to sevoflurane and has been used by two of the authors in private practice (Drs. Troyer and Feeman) with good success. A longer induction and recovery period would be expected with isoflurane, as would a lower induction and maintenance dose.
H.L. Troyer, DVM*
William E. Feeman III, DVM**
Toni L. Gray, RVT***
C. Guillermo Couto, DVM, DACVIM
Department of Veterinary Clinical Sciences
College of Veterinary Medicine
The Ohio State University
Columbus, OH 43210
Current addresses:
*Oradell Animal Hospital
580 Winters Ave.
Paramus, NJ 07652
**Animal Medical Centre of Medina
1060 S. Court St.
Medina, OH 44256
***5230 Renner Road
Columbus, OH 43228
Supported in part by grant P30 CA16058, National Cancer Institute, Bethesda, Md.
REFERENCES
1. Muir WW, Hubbell JAE, Skarda RT, et al. Handbook of veterinary anesthesia. 3rd ed. St. Louis, Mo: Mosby Inc., 2000.
2. Plumb DC. Veterinary drug handbook. 4th ed. Ames: Iowa State Press, 2002.
3. Aronson CE. Veterinary pharmaceuticals and biologicals 1982-1983. Edwardsville, Kan: Veterinary Medicine Publishing Co., 1983.
4. USP DI—Drug information for the health care professional. Vol. 1, 17th ed. Rockville, Md: The United States Pharmacopeial Convention Inc., 1997.
5. Gaertner DJ, Boschert KR, Schoeb TR. Muscle necrosis in Syrian hamsters resulting from intramuscular injections of ketamine and xylazine. Lab Anim Sci 1987;37:80-83.
6. Smiler KL, Stein S, Hrapkiewicz KL, et al. Tissue response to intramuscular and intraperitoneal injections of ketamine and xylazine in rats. Lab Anim Sci 1990;40:60-64.
7. Budavari S. The Merck index—Encyclopedia of chemicals, drugs and biologicals. Rahway, NJ: Merck and Co. Inc., 1989.
8. McEvoy GK. American Hospital Formulary Service —Drug information 92. Bethesda, Md: American Society of Hospital Pharmacists Inc., 1992.
9. AMA drug evaluations annual. Chicago, Ill: American Medical Association, 1991.
10. Rall TW, Taylor P, Nies AS, et al. Goodman & Gilman's the pharmacological basis of therapeutics. 8th ed. New York, NY: McGraw-Hill, 1990.
11. Cullen LK. Medetomidine sedation in dogs and cats: a review of its pharmacology, antagonism and dose. Br Vet J 1996;152:519-35.
12. Dobromylskyj P. Cardiovascular changes associated with anaesthesia induced by medetomidine combined with ketamine in cats. J Small Anim Pract 1996;37:169-172.
13. Verstegen J, Fargetton X, Donnay I, et al. An evaluation of medetomidine/ketamine and other drug combinations for anaesthesia in cats. Vet Rec 1991;128:32-35.
14. Verstegen J, Fargetton X, Donnay I, et al. Comparison of the clinical utility of medetomidine/ketamine and xylazine/ketamine combinations for the ovariectomy of cats. Vet Rec 1990;127:424-426.
15. Vaha-Vahe T. Clinical evaluation of medetomidine, a novel sedative and analgesic drug for dogs and cats. Acta Vet Scand 1989;30:267-273.
16. Verstegen J, Fargetton X, Ectors F. Medetomidine/ ketamine anaesthesia in cats. Acta Vet Scand Suppl 1989;85:117-123.
17. Young CJ, Apfelbaum JL. Inhalational anesthetics: desflurane and sevoflurane. J Clin Anesth 1995;7:564-577.
18. Clarke KW. Desflurane and sevoflurane. New volatile anesthetic agents. Vet Clin North Am Small Anim Pract 1999;29:793-810.
19. Hikasa Y, Kawanabe H, Takase K, et al. Comparisons of sevoflurane, isoflurane, and halothane anesthesia in spontaneously breathing cats. Vet Surg 1996;25:234-243.