Cranial cruciate ligament injuries in dogswhere to go next?
Try this overview of surgical repair options to brush up your knowledge.
Canine cranial cruciate ligament (CCL) disease is the most common cause of hindlimb lameness seen in our canine patient population.1 Rupture of the ligament may occur as a traumatic avulsion, an acute tear from excessive strain or more commonly as a progressive degenerative disease resulting in partial or complete ligament rupture. While any dog breed can suffer from the condition, there are certain breeds that appear to be predisposed. Patients may present at any age, and both neutered and female patients may be over-represented.2, 3 Timing of neuter has not been shown to correlate with this disease, and the disease is thought to be multifactorial. We understand certain genetic, developmental and environmental factors play a role in the degenerative process, including limb anatomic conformation, tibial plateau angle, body condition and activity type, but all factors have not yet been identified. Since the degenerative form of the disease is thought to be progressive in nature, patients may present at varying ages and stages of ligament integrity. It has also been stated that 30% to 40% of patients who tear one cruciate ligament are predisposed to tearing the contralateral limb within one or two years.4
The cranial cruciate ligament is important to neutralizing cranial tibial subluxation, internal rotation and hyperextension of the stifle. The ligament itself consists of two bands: craniomedial and caudolateral. The craniomedial band is taut in both flexion and extension, while the caudolateral band is only taut in extension. For this reason, the craniomedial band is often the first portion of the ligament to fail.1 These patients are considered to have a partial tear, and medical management of these cases has shown variable success depending on the techniques used.
Diagnosing a CCL tear
High suspicion of partial or complete CCL tear is typically identified on physical examination coupled with two-view stifle radiographs. The most common physical exam findings include stifle joint effusion, medial buttress (with chronicity) along with cranial drawer sign and tibial thrust. Cranial drawer sign may only be present on flexion of the limb in patients with a partial CCL tear. These patients also tend to be painful on hyperextension of the stifle. Radiographs will typically confirm evidence of joint effusion and may show signs of osteoarthritis in the joint. Some patients may also show the tibia cranially displaced (in drawer) when compared with the femur (Figure 1).
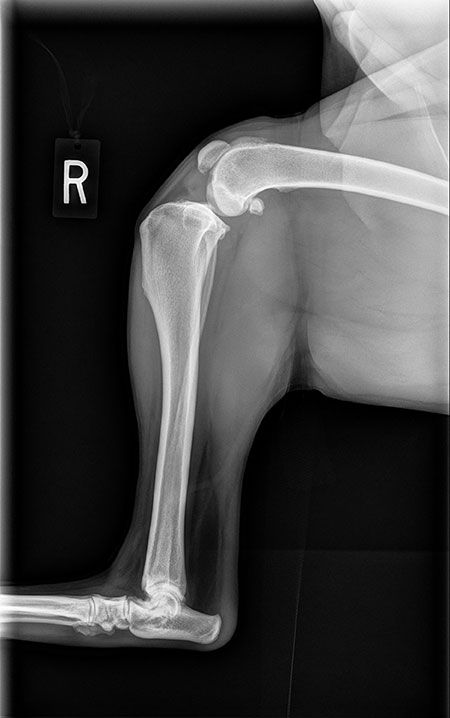
Figure 1: Right stifle, lateral view. The infrapatellar fat pad is reduced as a result of joint effusion and or capsular thickening. Mild osteophytes are seen on the distal femur, patella and tibial condyles. | All images courtesy of Dr. William Snell
Magnetic resonance imaging (MRI) is an uncommon diagnostic tool used for identification of CCL tears or meniscal disease in veterinary medicine at this time. Minimally invasive procedures including standard stifle arthroscopy or, more recently, arthroscopy via needle scope may also be used as a diagnostic tool to confirm the diagnosis of a partial or complete CCL tear.
Non-surgical management
Medical management options for partial CCL tears may consist of varying combinations of rest, nonsteroidal anti-inflammatory drugs (NSAIDs), nutraceutical use and physical therapy. But more recently platelet-rich plasma (PRP) and/or canine stem cells derived from bone or fat have been used with varying and, unfortunately, inconsistent success.5 There's also an argument that early surgical intervention in patients with partial tears may help in preserving the remainder of the intact ligament and that these patients may have a better postoperative success.
Surgical management options
Surgical options for these patients are broken down into two general categories: ligament replacement versus biomechanical techniques. More recent literature has suggested that using a combination of a ligament replacement and biomechanical techniques may result in a more stable stifle joint postoperatively.6 Ligament replacement techniques can be further subdivided into intra-articular or extra-articular techniques.
Regardless of stabilization procedure, it's routine to first perform a joint exploration. This may be achieved by an open arthrotomy or arthroscopically. All intra-articular structures should be evaluated, with special attention paid to the articular surfaces of the femur, tibia and patella as well as the long digital extensor tendon, both the cranial and caudal cruciate ligaments and the medial and lateral menisci. Meniscal tears should be débrided if present. There's also continued debate over the procedure known as a “meniscal release.” This technique involves transection of the caudal attachments of the medial meniscus with the goal of reducing the risk of subsequent meniscal tearing. However, it has been shown to result an increased contact area of the medial joint compartment, contributing to accelerated development of osteoarthritis in the stifle joint.7
Intra-articular stabilization techniques are the mainstay of human anterior cruciate ligament (ACL) repairs. These techniques employ harvesting a biologic graft from another tendon in the body or from a cadaver and then drilling bone tunnels and replacing the torn ACL. The graft will then go through a period of devitalization, followed by revascularization and ligamentization over an approximately 20-week period. During this time, the graft is at a diminished tensile strength, and overuse may lead to graft failure and returned stifle instability. More recent research has focused on synthetic intra-articular replacement or biologic scaffolds as opposed to the biological replacement techniques that have fallen out of favor in the veterinary surgical field. Today, however, most surgeons are finding themselves electing extra-articular or biomechanical stabilization techniques.
Extra-articular repair techniques utilize synthetic materials to traverse and stabilize the stifle joint. The most commonly performed techniques include the lateral fabellar suture, bone anchor techniques and the Tightrope technique. In all cases, material is anchored at relative isometric points outside the joint on the distal femur and proximal tibia. Given that there are no true isometric points in the stifle due to the cam-shaped femoral condyles, the synthetic material is subjected to cyclic loading that eventually results in implant fatigue and failure. The goal of these repair options is to stabilize the stifle long enough to allow for periarticular fibrotic tissue to develop and mature over a 16-week period. This periarticular fibrosis becomes the long-term stabilizer of the stifle joint. The main complications of these techniques include implant infection along with premature implant failure.8
Biomechanical techniques, also known as osteotomy procedures, involve cutting and manipulating the tibia in various ways that result in biomechanical neutralization of cranial tibial thrust in the CCL-deficient stifle. The most common procedures used today are the tibial plateau leveling osteotomy (TPLO), the tibial tuberosity advancement (TTA) and, more recently, the center of rotation angulation (CORA)-based leveling osteotomy (CBLO). Of these, the TPLO is probably the most widely used osteotomy technique.
With the TPLO procedure (Figure 2) the 90-degree flexed lateral radiograph of the tibia (including stifle and hock) allows for measurement of the tibial mechanical axis, and a bisecting joint line of the stifle allows for measurement of the needed tibial plateau angle (TPA). Average TPAs in dogs range from 25 to 30 degrees.9 Once the patient's TPA is measured, a dome osteotomy of the proximal tibia is performed and the proximal tibial fragment is rotated a predetermined distance based on the patient's specific TPA. This results in a reduction of the tibia slope to approximately five degrees, thus neutralizing cranial tibial thrust. A bone plate with screws is then used to bridge and stabilize the fracture on the medial tibia. The patient is then limited in activity over the next six to eight weeks while the osteotomy heals.
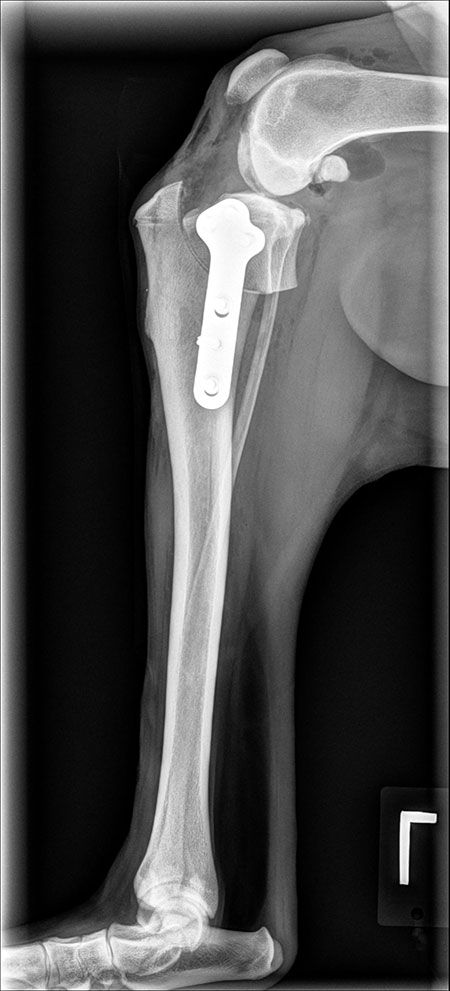
Figure 2: Left stifle, lateral view. Immediate postoperative TPLO.
The TTA (Figure 3) is a technique that involves a linear cut in the tibial tuberosity and advancement (cranial displacement) of the tibial tuberosity fragment in a cranial direction to achieve and maintain a patellar tendon angle of approximately 90 degrees with respect to the premeasured tibiofemoral contact point when the limb is in near full extension. This will in turn result in neutralization of the tibiofemoral shear force in a CCL-deficient stifle joint. The implant system consists of a cage and a bone plate that acts as a tension band. The cage is inserted within the proximal portion of the osteotomy and is size-specific to the intended cranial advancement of the tibial tuberosity fragment. The plate is then secured proximally to the medial aspect of the advanced tibial tuberosity fragment and distally to the tibial diaphysis acting as a tension band. The patient is then limited in activity over the next six to eight weeks while the osteotomy gap fills in and heals. Case selection is important with this technique, as specific tibial tuberosity conformations and excessive TPAs (> 30 degrees) may result in less favorable outcomes.
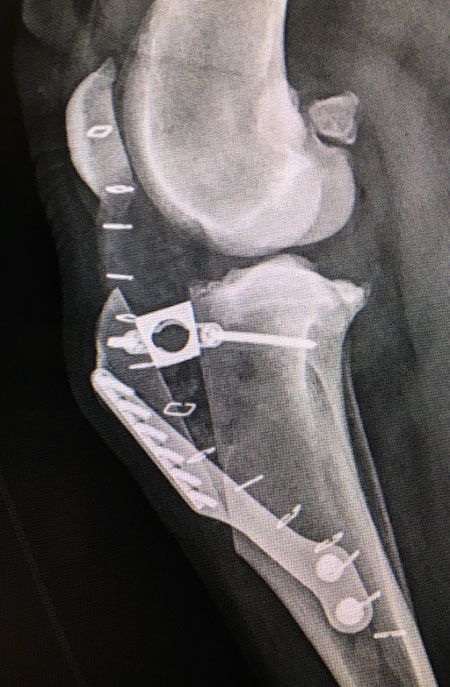
Figure 3: Left stifle, lateral view. Immediate postoperative TTA.
The CBLO (Figure 4) procedure is the most recently described procedure and is similar to the TPLO in that it is a dome osteotomy technique that attempts to level the TPA. The location of the osteotomy is based on the CORA of the tibia and attempts to place the axis of correction (ACA) in line with the CORA to limit tibial translation, thus limiting a caudal shift in joint forces that have been described with the TPLO procedure. An additional advantage over the TPLO includes the fact that it can be performed in juvenile patients, as the osteotomy and plate placement do not affect the proximal tibial physis and tibial tuberosity apophysis. Further advantages include an increase in fracture contact area for more load sharing, increased proximal fragment bone stock for additional implant placement and ease of adding on ancillary stabilizing procedures.
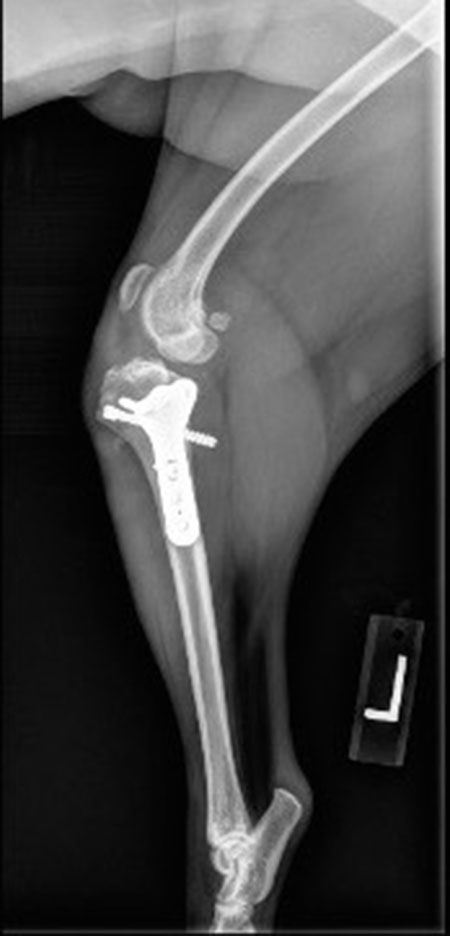
Figure 4: Left stifle, lateral view. Six weeks post-CBLO.
While each osteotomy procedure may have its own subset of complications specific to each individual technique, all procedures pose similar risks and complications of implant infection and/or failure. At the end of the day the procedure or procedures that are recommended are often determined based on the age, size, body condition of the patient, concomitant morbidities, the comfort level of the surgeon with the described techniques, the expectation of the owner and the pet owner's ability to financially support the recommended procedure.
Dr. William Snell is a board-certified small animal surgeon and is currently the medical director at Blue Pearl Veterinary Partners in Boston.
References:
1. “Stifle Joint.” Veterinary Surgery: Small Animal, by Karen M. Tobias, Elsevier Saunders, 2012, p. 906-972.
2. Duval JM, Budsberg SC, Flo GL, et al. Breed, sex, and body weight as risk factors for rupture of the cranial cruciate ligament in young dogs. J Am Vet Med Assoc. 1999 Sep 15;215(6):811-814.
3. Whitehair JG, Vasseur PB, Willits NH. Epidemiology of cranial cruciate ligament rupture in dogs. J Am Vet Med Assoc. 1993 Oct 1;203(7):1016-1019.
4. Doverspike M, Vasseur PB, Harb MF, et al. Contralateral cranial cruciate ligament rupture: incidence in 114 dogs. J Am Anim Hosp Assoc. 1993 29:167.
5. Canapp SO, Leasure CS, Cox C, et al. Partial cranial cruciate ligament tears treated with stem cell and platelet-rich plasma combination therapy in 36 dogs: A retrospective study. Front Vet Sci. 2016 Dec 14;3:112.
6. Schaible M, Shani J, Caceres A, et al. Combined tibial plateau levelling osteotomy and lateral fabellotibial suture for cranial cruciate ligament rupture with severe rotational instability in dogs, J Small Anim Pract. 2017 Apr;58(4):219-222.
7. Pozzi A, Kim S, Lewis D. Effect of transection of the caudal menisco-tibial ligament on femorotibial contact mechanics. Vet Surg. 2010 Jun;39(4):489-495.
8. Casale SA, McCarthy RJ. Complications associated with lateral fabellotibial suture surgery for cranial cruciate ligament injury in dogs: 363 cases (1997–2005). J Am Vet Med Assoc. 2009 Jan 15;234(2):229-235.
9. Griffon DJ. A Review of the pathogenesis of canine cranial cruciate ligament disease as a basis for future preventive strategies. Vet Surg. 2010 Jun;39(4):399-409.