Epsilon aminocaproic acid: A new tool for treating bleeding dogs
Bleeding people are commonly treated with antifibrinolytic agents, but these drugs are relatively uncommon in veterinary medicine. Learn more about their benefits-and potential drawbacks-so you can decide if they should be added to your clinical toolbox.
(shutterstock.com)Hemorrhage can be a frustrating and life-threatening clinical problem. Bleeding may result from trauma, surgical procedures, abdominal masses or coagulation abnormalities. Common techniques used to treat a bleeding patient include surgery to ligate the bleeding vessel, bandages and blood products to treat anemia. In human medicine, antifibrinolytic agents are commonly administered in combination with these techniques to treat bleeding patients. Treating veterinary patients with antifibrinolytic agents is uncommon but the beneficial effects of this therapy are being investigated.
Mechanism of action
The body's ability to form stable blood clots is vital to the control of bleeding (hemostasis). When a blood vessel is damaged, platelets and coagulation factors are activated, which ultimately produces a stable blood clot that slows down or stops the active bleed. These clots are then slowly broken down by a process called fibrinolysis. This process is important for restoring the lumen of the vessel and normalizing blood flow after an insult. Delayed fibrinolysis can result in a persistent clot and thromboembolic disease, which can have severe negative consequences for the patient.
Fibrinolysis occurs when plasminogen is converted to plasmin.1 Plasmin initiates the disintegration of the fibrin meshwork in clots, resulting in the breakdown of the clot (Figure 1). Excessive fibrinolysis may lead to accelerated clot breakdown, inadequate hemostasis and continued bleeding. Antifibrinolytic drugs help decrease the rate of fibrinolysis by slowing clot breakdown, thus helping to control hemorrhage.
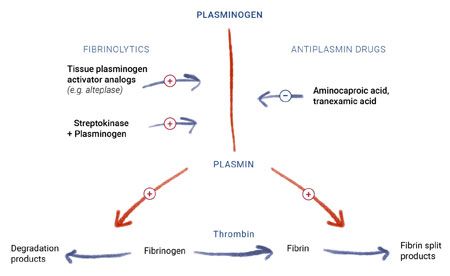
Figure 1. A graphic depiction of fibrinolysis. Adapted from: Trevor AJ, Katzung BG, Kruidering-Hall. Katzung & Trevor's Pharmacology: Examination and board review. 11th Ed. New York: McGraw-Hill Education, 2015.
Efficacy of antifibrinolytic drugs
The most commonly administered class of antifibrinolytic agents is synthetic lysine analogue, which includes aminocaproic acid, also known as epsilon aminocaproic acid (EACA), and tranexamic acid (TXA). EACA and TXA block the lysine binding site of plasminogen, which inhibits plasmin activation.2 The result is increased clot duration, which may be beneficial in numerous clinical conditions in which hyperfibrinolysis is thought to occur, including trauma, spontaneous hemoabdomen, neoplasia, liver disease and surgery.3-7 Hyperfibrinolysis may lead to increased morbidity and mortality as a result of uncontrolled bleeding.8
Interestingly, dogs may potentially be relatively hyperfibirinolytic when compared with people.9 Dogs have been shown to have increased euglobulin lysis time10 and increased plasma concentrations of tissue plasminogen activator,11 which aids in clot breakdown. Experimental dog models of pulmonary thromboembolism showed resolution of clinical signs within days versus within years in people.12
The effects of antifibrinolytic drugs are best evaluated with thromboelastography (TEG). TEG assesses the entire coagulation cascade, including clot formation and fibrinolysis, and displays it graphically (Figure 2).
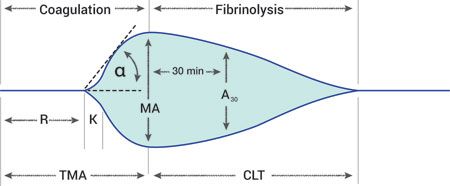
Figure 2. A TEG tracing. (Courtesy of Dr. Jason Couto)Standard coagulation assays, such as prothrombin time (PT) and partial thromboplastin time (PTT), do not evaluate the fibrinolytic phase of coagulation. Patients with hyperfibrinolysis may have a normal PT or PTT and may still continue to have severe hemorrhage that is unresponsive to the administration of blood products and treatment of the underlying cause. These patients are suspected to have clots that form normally but break down too quickly, which can lead to persistent bleeding. Administration of antifibrinolytic drugs in these dogs has been shown through TEG to increase clot strength (the MA value) and decrease clot lysis.6 Tissue plasminogen activator (tPA) catalyzes the conversion of plasmin to plasminogen and is used clinically in cases of thromboembolic disease to break down clots.13 When available, tPA-TEG may help predict the need for massive blood transfusions in human trauma and may be a useful tool for veterinarians monitoring trauma patients.14
TEG samples have to be run within 30 minutes of collection and are, thus, only practically useful for practitioners who have TEG within their clinical practice. However, even in the absence of advanced diagnostic tools, practitioners can monitor the rate and severity of clinical bleeding with close monitoring of the packed cell volume or hematocrit when administering antifibrinolytic drugs. Elevated lactate concentrations have also been associated with increased fibrinolysis and can be useful in indicating ongoing hyperfibrinolysis when TEG is not available.4
Indications and use
Antifibrinolytic agents have been administered to decrease blood loss in people with normal and increased fibrinolysis.2 These agents are given to people undergoing orthopedic, cardiac, pediatric and organ transplant surgeries to decrease postoperative bleeding and minimize blood transfusions. They are also given to patients with trauma or coagulopathies with active bleeding. Besides surgery and trauma, antifibrinolytic drugs have also been given to treat people with hematologic disorders, postpartum refractory bleeding, or upper gastrointestinal hemorrhage.15-17 A multicenter, randomized clinical trial (CRASH-2) has shown TXA's efficacy in decreasing overall mortality in human acute trauma,8 and a similar clinical trial (CRASH-3) for the treatment of traumatic brain injury is ongoing.18 Lysine analogues have found a clear, beneficial role in human medicine as hemostatic agents.2
A study showed that EACA (1 drop of 35.7 mg/ml solution in both eyes t.i.d.) was efficacious in treating nonhealing corneal ulcers in dogs because of its antiplasmin activity.19
Antifibrinolytics in dogs
While there is limited evidence evaluating the safety and efficacy of antifibrinolytics in veterinary medicine, preoperative EACA administration has been shown to decrease clinical postoperative bleeding in greyhounds.20 In one study, greyhounds with appendicular tumors that underwent amputation were 5.7 times more likely to bleed if they did not receive EACA than if they did.21
EACA is well-tolerated in dogs with clinical bleeding at a wide range of doses with minimal side effects.22 In one patient with presumed acute traumatic coagulopathy, EACA administration stabilized clot formation, which resulted in rapid clinical improvement.6 Results from these studies are promising and suggest a beneficial role for lysine analogues in patients with refractory bleeding and to potentially decrease surgery-associated blood loss.
Most of the studies and clinical use of antifibrinolytic drugs in veterinary medicine involve EACA because it has shown to be similar in efficacy to TXA but is a fraction of the price.9 The injectable form of EACA is significantly less expensive than the oral tablet form is, and the injectable form can be given orally safely and with similar efficacy without adverse effect in dogs.23 The same study also suggested that a dose of 100 mg/kg results in increased duration of fibrinolysis inhibition when compared with a 20 mg/kg dose. There were no adverse effects associated with any doses in that study.23
Antifibrinolytics in cats
To our knowledge, there are no studies in the veterinary literature investigating the role of lysine analogues in bleeding cats. There is limited published information on fibrinolysis in cats.
Recommendations for practitioners
EACA appears to be beneficial in treating hemorrhage in dogs. Below are our recommendations for administering this drug in your practice.
Greyhounds undergoing surgery
Despite having an appropriate number of functional platelets and normal clotting times, up to one-third of greyhounds experience profound postoperative or trauma-associated bleeding, even after minor surgical procedures.24 This clinical syndrome can become life-threatening, especially given the high frequency of limb amputation due to greyhounds' predisposition for appendicular osteosarcoma. The administration of EACA (500 mg orally t.i.d.) on the day of surgery and for four days after surgery significantly decreased postoperative bleeding in greyhounds.20,21
We recommend routine administration of EACA in greyhounds before any elective or emergent surgical procedure, and that is a routine practice in our institution. We recommend an EACA dosage of 50 to 100 mg/kg orally or intravenously every six hours.
Active bleeding
EACA has been given to dogs with clinical conditions that include pericardial effusion,25 hemoabdomen, liver failure,3 bleeding subcutaneous masses and epistaxis. At our institution, we routinely administer EACA for these clinical conditions to limit the giving of blood products and to control blood loss. In one study, the dose of EACA ranged from 9 to 111 mg/kg with minimal side effects noted.19
EACA can be given at the hospital to patients with acute blood loss, but it can also be given at home in applicable cases (e.g. dogs with pericardial effusion).
One study showed that the routine administration of EACA and Yunnan Baiyao (a traditional Chinese hemostatic medication) to dogs with right atrial masses and pericardial effusion was safe.25 However, in this study, EACA administration did not lead to improved survival times. The median dose of EACA used in this study was 48.8 mg/kg, and it was given three to four times a day.
In dogs with active bleeding, we recommend a dosage of 50 to 100 mg/kg orally or intravenously every six hours.9,23
Adverse effects
Lysine analogue therapy is not without consequence. Several studies have hinted that thromboembolic disease, specifically deep vein thrombosis, and seizures are potentially serious side effects in people receiving with TXA.26,27 Neither of these side effects have been documented in veterinary patients although diarrhea, decreased appetite and loss of multiple toe nails have been rarely reported in dogs receiving EACA.22 Although EACA appears to be safe at published dosages, more clinical research is needed to determine the adverse event profile of lysine analogue therapy.
Conclusion
Antifibrinolytic drugs appear to be a safe and relatively inexpensive therapeutic option for controlling hemorrhage in veterinary patients. While the evidence for beneficial preoperative antifibrinolytic therapy in human medicine is overwhelming, 2 more research is needed to determine its efficacy in veterinary patients.
References
1. Al-Horani RA, Desai UR. Recent advances on plasmin inhibitors for the treatment of fibrinolysis-related disorders. Med Res Rev 2014;34(6):1168-1216.
2. McCormack PL. Tranexamic acid: a review of its use in the treatment of hyperfibrinolysis. Drugs 2012;72(5):585-617.
3. Kelley D, Lester C, Shaw S, et al. Thromboelastographic evaluation of dogs with acute liver disease. J Vet Intern Med 2015;29(4):1053-1062.
4. Fletcher DJ, Rozanski EA, Brainard BM, et al. Assessment of the relationships among coagulopathy, hyperfibrinolysis, plasma lactate, and protein C in dogs with spontaneous hemoperitoneum. J Vet Emerg Crit Care 2016;26(1):41-51.
5. da Luz LT, Nascimento B, Rizoli S. Thrombelastography (TEG): practical considerations on its clinical use in trauma resuscitation. Scand J Trauma Resusc Emerg Med 2013;21:29.
6. Yoo SH, Venn E, Sullivan LA, et al. Thromboelastographic evidence of inhibition of fibrinolysis after ε-aminocaproic acid administration in a dog with suspected acute traumatic coagulopathy. J Vet Emerg Crit Care 2016;26(5):737-742.
7. Kristensen AT, Wiinberg B, Jessen LR, et al. Evaluation of human recombinant tissue factor-activated thromboelastography in 49 dogs with neoplasia. J Vet Intern Med 2008;22(1):140-147.
8. CRASH-2 trial collaborators, Shakur H, Roberts I, et al. Effects of tranexamic acid on death, vascular occlusive events, and blood transfusion in trauma patients with significant haemorrhage (CRASH-2): a randomised, placebo-controlled trial. Lancet 2010;376(9734):23-32.
9. Fletcher DJ, Blackstock KJ, Epstein K, et al. Evaluation of tranexamic acid and ε-aminocaproic acid concentrations required to inhibit fibrinolysis in plasma of dogs and humans. Am J Vet Res 2014;75(8):731-738.
10. Cade JF, Robinson TF. Coagulation and fibrinolysis in the dog. Can J Comp Med 1975;39(3):296-298.
11. Lang IM, Marsh JJ, Konopka RG, et al. Factors contributing to increased vascular fibrinolytic activity in mongrel dogs. Circulation 1993;87(6):1990-2000.
12. Moser KM, Cantor JP, Olman M, et al. Chronic pulmonary thromboembolism in dogs treated with tranexamic acid. Circulation 1991;83(4):1371-1379.
13. Hooi KS, Lemetayer JD. The use of intravesicular alteplase for thrombolysis in a dog with urinary bladder thrombi. J Vet Emerg Crit Care 2017;27(5):590-595.
14. Moore HB, Moore EE, Chapman MP, et al. Viscoelastic tissue plasminogen activator challenge predicts massive transfusion in 15 minutes. J Am Coll Surg 2017;225(1):138-147.
15. Marshall A, Li A, Drucker A, et al. Aminocaproic acid use in hospitalized patients with hematological malignancy: a case series. Hematol Oncol 2016;34(3):147-153.
16. Wang HY, Hong SK, Duan Y, et al. Tranexamic acid and blood loss during and after cesarean section: a meta-analysis. J Perinatol 2015;35(10):818-825.
17. Gluud LL, Klingenberg SL, Langholz SE. Systematic review: tranexamic acid for upper gastrointestinal bleeding. Aliment Pharmacol Ther 2008;27(9):752-758.
18. Dewan Y, Komolafe EO, Mejía-Mantilla JH, et al. CRASH-3-tranexamic acid for the treatment of significant traumatic brain injury: study protocol for an international randomized, double-blind, placebo-controlled trial. Trials 2012;13:87.
19. Regnier A, Cazalot G, Cantaloube B. Topical treatment of non-healing corneal epithelial ulcers in dogs with aminocaproic acid. Vet Rec 2005;157(17):510-513.
20. Marín LM, Iazbik MC, Zaldivar-Lopez S, et al. Epsilon aminocaproic acid for the prevention of delayed postoperative bleeding in retired racing greyhounds undergoing gonadectomy. Vet Surg 2012;41(5):594-603.
21. Marín LM, Iazbik MC, Zaldivar-Lopez S, et al. Retrospective evaluation of the effectiveness of epsilon aminocaproic acid for the prevention of postamputation bleeding in retired racing Greyhounds with appendicular bone tumors: 46 cases (2003-2008). J Vet Emerg Crit Care 2012;22(3):332-340.
22. Davis M, Bracker K. Retrospective study of 122 dogs that were treated with the antifibrinolytic drug aminocaproic acid: 2010-2012. J Am Anim Hosp Assoc 2016;52(3):144-148.
23. Brown JC, Brainard BM, Fletcher DJ, et al. Effect of aminocaproic acid on clot strength and clot lysis of canine blood determined by use of an in vitro model of hyperfibrinolysis. Am J Vet Res 2016;77(11):1258-1265.
24. Feeman WE III. Idiosyncrasies in greyhounds that can affect their medical care. Vet Med 2005;100(8):592-600.
25. Murphy LA, Panek CM, Bianco D, et al. Use of Yunnan Baiyao and epsilon aminocaproic acid in dogs with right atrial masses and pericardial effusion. J Vet Emerg Crit Care 2017;27(1):121-126.
26. Harvey R, Salehi A. Con: antifibrinolytics should not be used routinely in low-risk cardiac surgery. J Cardiothorac Vasc Anesth 2016;30(1):248-251.
27. Lecker I, Wang DS, Whissell PD, et al. Tranexamic acid-associated seizures: causes and treatment. Ann Neurol 2016;79(1):18-26.
Jason Couto, DVM, MS
Department of Small Animal Clinical Sciences
College of Veterinary Medicine
Michigan State University
Adesola Odunayo, DVM, MS, DACVECC
Department of Small Animal Clinical Sciences
College of Veterinary Medicine
University of Tennessee