Feline injection-site sarcomas update
Though still often called vaccine-associated fibrosarcoma, it is now known that many types of injections can cause this aggressive form of neoplasia in cats. Monitoring injection sites is key to a good prognosis, as is an optimal treatment protocol.
Feline injection-site sarcomas (ISSs) are distinct from soft tissue sarcomas that develop unrelated to injections because of their etiology and extremely aggressive invasion into surrounding tissues. Although ISSs have an overall low incidence, achieving disease control can be challenging. And while ISSs were initially described at the sites of vaccines, other reported causes of ISSs include injection sites of microchips, long-acting antibiotics, long-acting glucocorticoids and lufenuron, plus reaction sites to nonabsorbable sutures.1
Initial preventive measures
Given the need for wide surgical margins and subsequent difficulty in achieving control of interscapular ISS with surgery, the Vaccine-Associated Feline Sarcoma Task Force (VAFSTF) published the following recommendations in 1999 for vaccine administration2:
- Rabies vaccine: Right rear limb, as distal as possible
- Feline leukemia virus (FeLV) vaccine: Left rear limb, as distal as possible
- Other vaccinations: Right forelimb, as distal as possible
Since publication of these recommendations, the prevalence of ISSs has not changed,3 but diagnosis of ISS in the subcutaneous tissue of the pelvic limbs, lateral abdomen and thoracic limbs has increased while ISS in the interscapular region has decreased. Of particular note is the increase in lateral abdominal wall ISSs, which is hypothesized to be due to aberrant injection (vaccination intended for the pelvic limb).4
The VAFSTF also recommends that veterinarians and clients monitor any vaccine site, being sure to investigate when a lesion meets any one of the following criteria, called the 3-2-1 rule2:
- Persistence after 3 months post-injection
- Size larger than 2 cm at any time point
- Increase in size 1 month following injection
Investigation should include either Tru-Cut or wedge biopsy, ensuring that any subsequent surgery is not extended by the biopsy location and technique. Marginal excision and excisional biopsy are not appropriate for ISSs.
Staging
A minimum database is useful to assess a patient's overall health. Three-view thoracic radiographs are also an important staging tool, as most ISSs are high-grade sarcomas with a reported metastatic rate of up to 25%, most commonly to the lungs. Metastasis to the subcutaneous tissue, liver and lymph nodes has also been reported, so abdominal imaging-ultrasound or computed tomography (CT)-is appropriate in some cases.1-5
While soft tissue sarcomas are considered locally invasive tumors in general, ISSs are extremely invasive. Often, the mass palpable on physical examination represents only a small portion of the tumor itself. Advanced imaging using CT or magnetic resonance imaging (MRI) is essential in all cases of ISS to determine the extent of invasion and plan adequate surgical margins (Figures 1 and 2).5
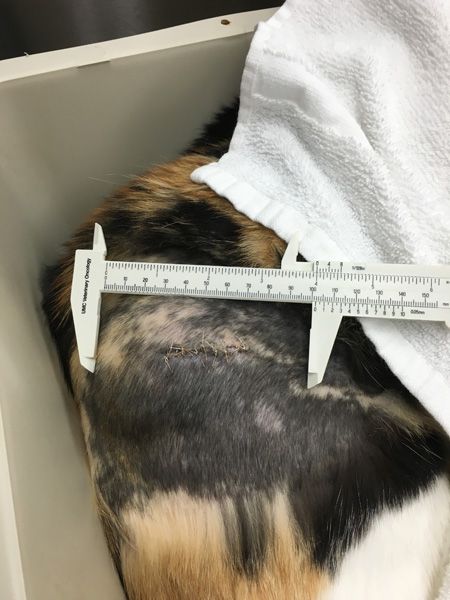
Figure 1. An injection site sarcoma of the proximal right thigh after an incisional biopsy. (Figures courtesy of Dr. Sandra Bechtel)
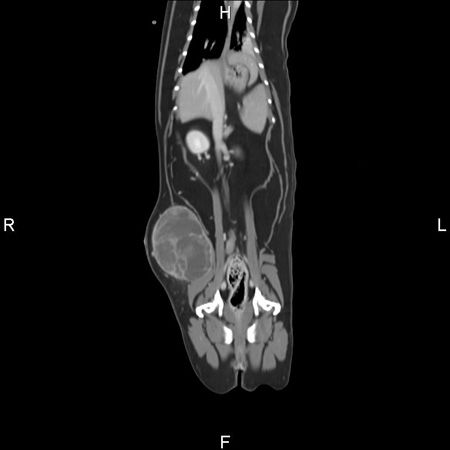
Figure 2. A contrast CT image of the cat in Figure 1. The coronal view revealed a moderately circumscribed and irregular mass extending into the abdominal wall.
Treatment
Surgery. ISSs are poorly encapsulated and extend along and infiltrate fascial planes, so referral with advanced imaging is optimal. When measured with contrast CT, tumor volume is approximately double the measurements from physical examination.5 If marginal resection is performed, the median time to recurrence is 66 to 79 days, with recurrence rates over 69%. However, when radical excision involving 5-cm margins without further therapy was performed in one retrospective study, the recurrence rates were 14%. In this same study, the median survival time in cats without recurrence was 1,461 days, while the median survival time in cats with recurrence was 499 days.6,7 This underscores the importance of an aggressive resection by a board-certified surgeon, as this offers the best chance for local control of ISSs. It is also important to note that radical surgical excision at the time of the first surgery imparts a better prognosis (improved disease-free intervals and survival times) compared with radical excision for recurrent tumors.8
The location of ISSs plays an important role in the ability to achieve tumor control. ISSs of the distal limb, for example, can be controlled in many cases by amputation. However, ISSs of the thoracic or abdominal wall or of the interscapular region require much more invasive surgeries to achieve the desired surgical margins.
Radiation. Radiation therapy is commonly used preoperatively or postoperatively to increase the disease-free interval in cats with ISSs, as incomplete tumor margins are common.9
Chemotherapy. The role of chemotherapy has not yet been defined and is generally reserved for cats in which surgery, radiation therapy or both are not available. The overall response rate to single-agent doxorubicin in cats with a palpable tumor is 33%10 and to single-agent ifosfamide is 41%.11 However, the median time to progression after administering either chemotherapeutic is short, ranging from 70 to 84 days. The role of chemotherapy in delaying or decreasing metastatic disease has yet to be defined.
Immunotherapy. Immunotherapy using a recombinant canary pox virus expressing feline interleukin-2 has been investigated for use in cats after surgery and radiation therapy, if the initial tumor was less than 5 cm.12 This therapy holds promise as an adjunctive treatment with the goal of improving local disease control.
The take-homes
Even though the incidence is low, ISSs are frustrating to treat and are often found when they are already large and difficult to control. And ISSs can be life limiting. Vaccinating according to VAFSTF guidelines can be helpful, in that cats developing ISSs may have a location more amenable to aggressive surgery (such as amputation), affording longer-term control. Increasing client awareness and implementing the 3-2-1 rule of monitoring post-vaccination may allow earlier detection and, therefore, improved results with therapy. Recommended treatment includes staging, advanced imaging and the first surgery being performed by a board-certified surgeon. Most cats with ISSs will also benefit from follow-up radiation therapy.
The role of chemotherapy is undefined. Immunotherapy with recombinant canary pox virus expressing feline interleukin-2 shows promise when used in combination with surgery and radiation therapy for small (less than 5 cm) ISSs. The most important aspects of treatment for ISSs are early identification of disease, advanced imaging and surgery at a referral institution.
References
1. Zabielska-Koczywas K, Wojtalewicz A, Lechowski R. Current knowledge on feline injection-site sarcoma. Acta Vet Scand 2017;59(1):47.
2. Vaccine-Associated Feline Sarcoma Task Force guidelines. Diagnosis and treatment of suspected sarcoma. J Am Vet Med Assoc 1999;214 (12):1745.
3. Wilcock B, Wilcock A, Bottoms K. Feline postvaccinal sarcoma: 20 years later. Can Vet J 2012;53(4):430-434.
4. Shaw SC, Kent MS, Gordon IK, et al. Temporal changes in characteristics of injection-site sarcomas in cats: 392 cases (1990-2006). J Am Vet Med Assoc 2009;234(3):376-380.
5. McEntee MC, Page RL. Feline vaccine-associated sarcomas. J Vet Intern Med 2001;15(3):176-182.
6. Phelps HA, Kuntz CA, Milner RJ, et al. Radical excision with five-centimeter margins for treatment of feline injection-site sarcomas: 91 cases (1998-2002). J Am Vet Med Assoc 2011;239(1):97-106.
7. Martin M. Vaccine-associated fibrosarcoma in a cat. Can Vet J 2003;44(8):660-663.
8. Müller N, Kessler M. Curative-intent radical en bloc resection using a minimum of a 3 cm margin in feline injection-site sarcomas: a retrospective analysis of 131 cases. J Feline Med Surg 2017. Epub ahead of print.
9. Martano M, Morello E, Buracco P. Feline injection-site sarcoma: past, present and future perspectives. Vet J 2011;188(2):136-141.
10. Poirier VJ, Thamm DH, Kurzman ID, et al. Liposome-encapsulated doxorubicin (Doxil) and doxorubicin in the treatment of vaccine-associated sarcoma in cats. J Vet Intern Med 2002;16:726-731.
11. Rassnick KM, Rodriguez CO, Khanna C, et al. Results of a phase II clinical trial on the use of ifosfamide for treatment of cats with vaccine-associated sarcomas. Am J Vet Res 2006;67(3):517-523.
12. Jourdier TM, Moste C, Bonnet MC, et al. Local immunotherapy feline fibrosarcomas using recombinant poxviruses expressing interleukin 2 (IL2). Gene Ther 2003;10(26):2126-2132.
Sandra Bechtel, DVM, DACVIM (oncology), is an associate professor of medical oncology at the University of Missouri's Veterinary Health Center. Her clinical interests include quality-of-life-focused cancer care and cancer diagnosis and therapy.