Medical therapies for canine pituitary-dependent hyperadrenocorticism
Hyperadrenocortocism is a common endocrinopathy in geriatric dogs.
HYPERADRENOCORTICISM is a common endocrinopathy in geriatric dogs. In about 85% of affected dogs, hyperadrenocorticism is secondary to excess secretion of ACTH from a benign tumor of the pituitary gland. Although people with pituitary-dependent hyperadrenocorticism (PDH) are often treated surgically, surgery remains efficacious yet impractical in dogs.1 Radiation therapy may slow the growth of pituitary tumors, but it often fails to control the hormonal effects of PDH.2,3 Thus, the mainstay of therapy in dogs with PDH is medical. The use of mitotane for selective adrenocorticolysis to treat PDH is well-described.4-8 This article discusses this use of mitotane and other medical therapies for treating PDH (Table 1).

Table 1 Clinically Useful Medical Therapies for Canine PDH
Mitotane therapy
In the late 1940s, it was noted that dogs given the insecticide dichloro-diphenyl-dichloroethane (DDD) developed severe adrenocortical necrosis and atrophy.9 The therapeutic use of mitotane, a derivative of DDD, for hyperadrenocorticism in dogs was described in 1973, and this form of therapy has gained widespread acceptance.10 A recent survey of more than 200 veterinary internists and dermatologists revealed that 95.6% preferred mitotane to other therapies for the treatment of PDH in dogs.11
Mechanism of action
Mitotane exerts its cortisol-lowering effects through progressive, selective necrosis of the adrenal zona fasciculata and zona reticularis. The zona glomerulosa is relatively resistant to mitotane's cytotoxic effects, so normal aldosterone secretion is usually maintained.10 Mitotane also interferes with steroid biosynthesis, primarily by inhibiting the 11β-hydroxylase and cholesterol side-chain cleavage enzymes (Figure 1).12
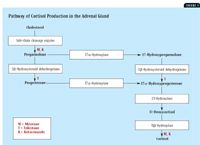
1. The enzymes involved in steroidogenesis are indicated in boxes. Specific steps in hormone synthesis blocked by the drugs mitotane ( M), trilostane (T), and ketoconazole (K) are indicated.
Mitotane is fat-soluble, distributed to virtually all tissues, and stored primarily in adipose tissue. It is converted to its active form by mitochondrial P-450 monooxygenases.13 Although the metabolic disposition of mitotane is unknown in dogs, mitotane likely undergoes oxidative metabolism in the liver as it does in people. This pathway may be clinically important, since the induction of hepatic microsomal enzymes by drugs such as phenobarbital may increase mitotane metabolism and, concurrently, diminish the adrenocorticolytic effect.12,14
Safety
Most of mitotane's adverse effects are related to the rapid decrease in serum cortisol concentrations or to hypocortisolemia itself.15 Lethargy, ataxia, weakness, anorexia, vomiting, or diarrhea occurs in about 25% of patients.14,16 These effects are usually mild and resolve with administration of glucocorticoids. Permanent hypoadrenocorticism (Addison's disease) develops in about 2% to 5% of dogs with PDH treated with the selective adrenocorticolysis mitotane protocol.12 Despite the relative resistance of the zona glomerulosa to mitotane's effects, hypoadrenocorticism may include hypoaldosteronism with resultant hyponatremia and hyperkalemia in addition to hypocortisolemia.4,5,17 Rarely, drug-induced central nervous system (CNS) signs occur weeks to months after maintenance therapy is initiated, including wandering, circling, and head pressing. Typically, these CNS signs are transient and are abolished by giving lower doses of mitotane more frequently.18 Hepatic changes, including congestion, centrilobular atrophy, and moderate to severe fatty degeneration, have been noted.16
Efficacy
Mitotane therapy is effective in 85% to 90% of dogs with PDH.4,6,8,10,15 Despite initial efficacy, about half of dogs treated with mitotane relapse within the first year of therapy.4,8,12 Patients that relapse usually respond to reinduction and an increased maintenance dosage.12
Administration and monitoring
Most clinicians still follow the mitotane protocol originally proposed for treating canine PDH in 1973.10 This protocol includes an induction period followed by long-term maintenance therapy. The induction dosage of mitotane for PDH is 30 to 50 mg/kg/day for eight to 10 days or until signs suggestive of hypoadrenocorticism develop.4,6,8,10,15 When possible, the daily dose should be divided into two equal doses and administered with food to increase absorption. Concurrent glucocorticoid supplementation with prednisone or prednisolone (0.15 to 0.25 mg/kg/day orally) can be used to mitigate the possible adverse effects associated with rapid serum cortisol concentration reduction.4,5,12 However, glucocorticoid supplementation during induction makes recognition of therapeutic end points difficult. If glucocorticoids are not concurrently administered, it is imperative that clients are provided with glucocorticoids in case signs of potentially life-threatening hypoadrenocorticism develop.12
At-home monitoring is crucial, especially during the induction period. Vomiting, weakness, anorexia, depression, or ataxia may be signs of mitotane overdosage, or they may be due to stomach upset associated with drug administration.14 Owners should be educated to recognize these warning signs and instructed to discontinue mitotane, administer oral prednisone, and consult their veterinarians if these signs occur. In the absence of these adverse reactions, mitotane should be discontinued when water consumption in a previously polydipsic dog decreases, when appetite wanes, or after the initial eight to 10 days of therapy. At that time, ACTH stimulation test results and serum electrolyte concentrations should be evaluated. Glucocorticoid therapy should not be given the morning of the ACTH stimulation test to avoid cross-reaction with the cortisol assay and falsely elevated cortisol concentrations.14
The goal of mitotane induction therapy is to achieve a basal cortisol concentration of 1 to 4 µg/dl with little or no increase in the cortisol concentration after ACTH stimulation.8,14 If the cortisol concentration is suppressed excessively, discontinuing mitotane for two to six weeks usually returns the serum cortisol concentration to normal.18 If the post-ACTH stimulation cortisol concentration remains elevated, induction may be continued for another five to seven days or until the previously mentioned clinical evidence of cortisol suppression is observed, whichever comes first. This course may be repeated if suppression is again inadequate. However, if suppression of cortisol has not been achieved by 21 days, the possibility of an adrenal tumor should be reconsidered.
Once pre- and post-ACTH stimulation cortisol concentrations are between 1 and 5 µg/dl, initiate maintenance therapy. Maintenance therapy involves dividing the daily mitotane induction dose (25 to 50 mg/kg) over two or three days of the week (e.g. Wednesday and Saturday or Monday, Wednesday, and Friday).6 Tablets are supplied in a 500-mg size, yet most dogs with PDH require much smaller doses. Because of the potential for toxicosis, owners should not crush or break the tablets themselves; trained personnel should formulate the tablets to the appropriate size. Be sure to tell owners to wear gloves or wash their hands after handling the medication.
Repeat serum electrolyte concentration measurements and ACTH stimulation testing in three and six months and then every six months thereafter. If the ACTH stimulation test results are above the desired ranges, increase the dose of mitotane by 25% to 50%.6 If a conservative increase in the mitotane dose does not alleviate clinical signs and decrease the cortisol concentrations appropriately within two months, repeat mitotane induction.6
ADDITIONAL THERAPIES
With greater insight into the pathophysiology of PDH, other therapies have been proposed. These therapies can be categorized based on mechanism: inhibitors of steroidogenesis (e.g. nonselective adrenocorticolysis with mitotane, ketoconazole, trilostane), central neuromodulators (e.g. L-deprenyl), and glucocorticoid-receptor antagonists (e.g. mifepristone). The following therapies may be useful in treating dogs with PDH (Table 1), but others cannot be recommended (Table 2).

Table 2 Alternative Proposed Medical Therapies for Canine PDH
Nonselective adrenocorticolysis with mitotane
In 1988, an alternative protocol for the medical management of PDH in dogs with mitotane was proposed.19 This protocol is popular in Europe because of the reduced long-term cost of mitotane and an increase in the disease-free interval.19-22 But the expense associated with proper monitoring and administration of mineralocorticoids may obviate any price differences.
Mechanism of action
This protocol delivers a higher cumulative initial dosage of mitotane with the goal of inducing complete, permanent destruction of the zona fasciculata and zona reticularis. In addition, concurrent lysis of the zona glomerulosa results in mineralocorticoid deficiency.4,5 Cortisone acetate and fludrocortisone acetate or desoxycorticosterone pivalate (Percorten-V—Novartis Animal Health) and sodium chloride are administered to ameliorate adrenocortical insufficiency (i.e. hypoadrenocorticism).14,20,21
Safety
This protocol preemptively treats for rapid decreases in serum cortisol concentrations and hypoaldosteronism, so dogs should experience few adverse effects if the medications are administered properly. When this protocol was first attempted in 49 dogs, transient adverse effects occurred in about 20% of dogs.19 Similar to dogs treated with selective adrenocortical lysis, only 4% to 6% of dogs developed severe signs of mineralocorticoid deficiency despite prescribed mineralocorticoid supplementation.6,19 In a separate study of 129 dogs, 30% developed clinical signs of hypoadrenocorticism requiring alteration in mitotane administration, but the clinical signs dissipated after withholding mitotane with no long-term complications.20
Whereas failure to administer maintenance mitotane poses no immediate life-threatening consequences, failure to administer glucocorticoids or mineralocorticoids to dogs treated with the nonselective adrenocortical lysis protocol may result in an addisonian crisis.4,5 For this reason, this protocol should be offered to only the most compliant owners. Strict adherence to medication administration and careful monitoring are necessary to prevent the potentially deadly complications of hypoadrenocorticism.
Efficacy
Nonselective adrenocortical lysis results in complete remission in more than 86% of dogs with PDH, with a median disease-free interval of 402 days.19,22 One study found that 26% of dogs required a second course of treatment within one year, but 29% of dogs were in remission for two years or longer.19 In another study, 77% of dogs were estimated to be free of disease based on owner observation of clinical signs after one year, 53% after two years, and 44% after three years.22 In contrast, about half of dogs treated with the selective adrenocortical lysis protocol will relapse within one year.6 Although it appears the nonselective protocol may have a longer disease-free interval, whether this benefit justifies the increased risks associated with hypoadrenocorticism is unknown. Noncompliance in the treatment of PDH is rarely life-threatening; noncompliance in the treatment of hypoadrenocorticism is life-threatening.
Administration and monitoring
Mitotane is administered orally with food at 50 to 75 mg/kg/day divided into three or four doses for 20 to 25 days.19-22 Lifelong replacement therapy with glucocorticoids and mineralocorticoids is begun on the third day. Described protocols involve the use of cortisone acetate at a temporarily high dosage of 2 mg/kg daily and fludrocortisone at 0.0125 mg/kg/day.21 After induction is complete, physical examination findings and serum electrolyte concentrations should be evaluated. If a dog is doing well clinically, the glucocorticoid dosage can be reduced to physiologic dosages (1 mg/kg/day of cortisone acetate).21 This protocol also advocates the oral administration of sodium chloride, but salt supplementation is often omitted from the treatment of hypoadrenocorticism without detriment.5,21 As an alternative to cortisone, prednisone or prednisolone may be used at an equipotent dosage of 0.4 mg/kg/day initially or 0.2 mg/kg/day after induction. Desoxycorticosterone (about 2.2 mg/kg every 25 days) could be used as a mineralocorticoid replacement alternative to fludrocortisone.19,22 Routine reevaluation should occur every four to six months.4,5,17,21,22
Trilostane
Trilostane is used throughout Europe and Asia as a safe, efficacious therapy for canine PDH. Although approved for use in people by the FDA, trilostane is no longer manufactured in the United States. With FDA permission, pet owners in the United States can obtain trilostane from several Internet or overseas pharmacies (see boxed text titled "Protocol for importing trilostane").

Protocol for importing trilostane
Mechanism of action
Trilostane is a synthetic, orally active steroid analogue. It acts as a competitive inhibitor of the 3β-hydroxysteroid dehydrogenase enzyme system, which blocks the production of corticosteroids, mineralocorticoids, and sex hormones (Figure 1).23 The effects of trilostane are largely reversible and are dose-dependent.24
Safety
Trilostane seems to be well-tolerated by dogs. Mild lethargy and decreased appetite are occasionally seen two to four days after initiating therapy, likely due to steroid withdrawal.24,25 Mild hyperkalemia, azotemia, hyperbilirubinemia, and hypercalcemia have been reported but were usually not associated with clinical illness.26 In one study, two of 78 dogs developed hypoadrenocorticism, while a second study involving 30 dogs reported no troubling side effects.25,26 Bilateral adrenal gland enlargement has been reported but was not clinically important.27
Efficacy
Trilostane seems to be highly effective in resolving the signs of PDH.26-28 Polyuria, polydipsia, and polyphagia were ameliorated in 70% of 67 dogs with these clinical signs.26 A marked improvement in dermatologic signs was noted in 62% of 39 dogs.26 Significant reductions in both basal and post-ACTH stimulation cortisol concentrations were observed after a mean of 12.3 days.26 Target cortisol concentrations were achieved in 81% of 73 dogs within the first month of therapy, with an additional 15% achieving target cortisol concentrations subsequently.26 Two other studies reported 82% to 97% efficacy based on resolution of clinical signs and normalization of ACTH stimulation test results.25,27
Administration and monitoring
Pharmacokinetic data for trilostane in dogs are not available, so the optimal dose and frequency interval for treating PDH are unknown. A typical starting dosage for dogs with PDH is 5 to 10 mg/kg given orally once a day, although some dogs respond better to twice-a-day dosing.24 In a study of 30 dogs, 19 mg/kg/day was the mean dosage required to maintain remission.25 Doses up to 40 to 50 mg/kg have been given with no adverse effects.28,29 Similar to mitotane, trilostane should be given with food to increase its absorption.
Careful monitoring is imperative to achieve optimal control of PDH. Perform ACTH stimulation testing and measure serum electrolyte concentrations 10 to 14 days, 30 days, and 90 days after initiating therapy. It is important to perform these tests four to six hours after trilostane administration. If the post-ACTH cortisol concentration is less than 1 µg/dl, discontinue the trilostane for 48 hours and then reintroduce it at a lower dose (generally the next lower capsule size).28 If the post-ACTH cortisol concentration is greater than 4.3 µg/dl, increase the dose of trilostane to the next higher capsule size.28 If the post-ACTH cortisol concentration is between 1 and 4.3 µg/dl, serum electrolyte concentrations are normal, and the dog appears clinically normal, keep the dose as is. After the dosage has been stabilized and the dog is free of clinical signs, ACTH stimulation test results and serum electrolyte concentrations should be monitored every three or four months.28
Ketoconazole
Ketoconazole, commonly used to treat fungal infections, is an imidazole drug that also inhibits steroidogenesis. The expense and twice-a-day dosing of ketoconazole have limited its routine use in treating PDH in dogs. Ketoconazole is more often used to treat dogs with mitotane-resistant adrenocortical tumors, particularly to stabilize patients before adrenalectomy.
Mechanism of action
Ketoconazole interferes with gonadal and adrenal steroid synthesis by inhibiting cytochrome P-450–dependent enzymes.16,30 It inhibits both the cholesterol side-chain cleavage step and the 11β-hydroxylation step (Figure 1).30 Other imidazole antifungal drugs, such as itraconazole, do not inhibit steroidogenesis.31
Safety
In a report of 18 dogs with PDH given ketoconazole, the only adverse effect noted was gastrointestinal upset in one dog.32 Hepatotoxicity has been reported, and thrombocytopenia is rarely encountered. Some dogs treated with ketoconazole will have a reversible lightening of their coats.16 The effects of hypocortisolemia are reversible with discontinuation of the drug, and mineralocorticoid deficiency is not observed.16
Efficacy
The efficacy of ketoconazole in dogs with PDH is debatable. A marked reduction in the serum cortisol concentration can occur within 30 minutes of administration and lasts from eight to 12 hours. In a single small study, 81% of dogs with PDH attained complete remission with ketoconazole.32 However, anecdotal evidence suggests the efficacy may be lower. In a survey of internists and dermatologists, most thought ketoconazole was effective in less than 25% of dogs with PDH.11 Only one of seven surveyed considered the efficacy of ketoconazole similar to that of mitotane.11 Treatment failure has been associated with poor bioavailability of orally administered ketoconazole.32 Assessing serum drug concentrations can differentiate between animals with poor intestinal absorption and those with true treatment failure.32
Administration and monitoring
Ketoconazole is administered initially at a dosage of 5 mg/kg given orally with a small meal twice a day for seven days. If no adverse effects occur, increase the dose to 10 mg/kg twice a day. After three weeks of therapy, perform ACTH stimulation testing within three to six hours of ketoconazole administration. If the basal and post-ACTH stimulation serum cortisol concentrations are not below 5 µg/dl and the clinical signs have not resolved, increase the dose by 50% again. Most dogs with PDH will not achieve remission with dosages below 30 mg/kg/day.4
L-Deprenyl
L-Deprenyl, or selegiline hydrochloride, is the only FDA-approved drug for treating PDH in dogs. It is also used to treat canine cognitive dysfunction. Its use for treating PDH is controversial.
Mechanism of action
L-Deprenyl's mechanism of action is irreversible inhibition of monoamine oxidase B, resulting in increased central dopamine concentrations. Several studies have indicated that dopamine can inhibit the release of ACTH by the pars intermedia and, possibly, the pars distalis.33-35 It is hypothesized that L-deprenyl, by increasing dopamine concentrations in the brain, inhibits ACTH secretion (Figure 2).6 Additionally, L-deprenyl may have a suppressive effect on the growth of pituitary adenomas, although more studies are warranted to confirm this.6
Safety
L-Deprenyl is generally quite safe in dogs. Studies to date have demonstrated few adverse effects in healthy dogs or dogs with PDH treated long-term with L-deprenyl.6,7,16,36,37 The most common adverse effects include vomiting, diarrhea, salivation, and anorexia. CNS effects can include restlessness, repetitive movements, or lethargy.16 Mineralocorticoid deficiency does not occur.
Efficacy
The efficacy of L-deprenyl for treating PDH remains controversial. Because L-deprenyl is neither adrenocorticolytic nor an inhibitor of steroidogenesis, ACTH stimulation testing cannot be used to monitor efficacy. Owners' impressions of improvement have been used to measure treatment efficacy in several studies. Because a metabolite of L-deprenyl is a potent sympathomimetic and CNS stimulant, perceived improvements in the behavior of geriatric dogs with PDH may not be directly attributable to control of PDH.38,39 Improvements in clinical signs of PDH in up to 77% of dogs treated with L-deprenyl have been reported in studies sponsored by the manufacturer.36,40 In these same studies, low-dose dexamethasone suppression test results normalized at one or more time points in only 17% of patients. These studies have been criticized for relying largely on subjective data collected from many independent observers.4
An independent investigation of L-deprenyl found that 20% of dogs with PDH exhibited a good response.38 This proportion is similar to the proportion of dogs with PDH resulting from a tumor of the pars intermedia rather than the more common pars distalis tumors (Figure 2). There is little evidence that dopamine plays a major role in suppressing ACTH secretion from the pars distalis, which may explain L-deprenyl's low efficacy in treating all dogs with PDH. Although dogs with pars intermedia tumors causing PDH may have a good response to L-deprenyl, no clinically applicable test exists to identify this subset of dogs.7 Because of its low efficacy, L-deprenyl is generally not recommended for treating PDH.4,5,7,24 In rare, select cases in which more effective treatments are contraindicated or when clinical signs are relatively mild, L-deprenyl therapy may be warranted.

2. Control of ACTH secretion from the pars intermedia portion of the pituitary gland differs from that of the pars distalis portion. Corticotrophs in the pars distalis are responsible for most ACTH secretion in health and for about 80% of all pituitary adenomas resulting in PDH. They are stimulated by both positive (corticotropin-releasing hormone) and negative (cortisol, ACTH) feedback. In the pars intermedia, serotoninergic and dopaminergic nerve fibers stimulate and suppress ACTH secretion, respectively. Theoretically, decreased serotonin concentrations (as induced by cyproheptadine) or increased dopamine concentrations (as induced by L-deprenyl or bromocriptine) should suppress ACTH secretion by the pars intermedia. Tumors of the pars intermedia are responsible for about 20% of dogs with PDH.
Administration and monitoring
L-Deprenyl is administered at a dosage of 1 mg/kg/day given orally, preferably in the morning. If no response is observed after two months of therapy, the dose can be doubled for an additional month. If a good response is seen, normalization of low-dose dexamethasone suppression test results and urinary cortisol:creatinine ratios should occur. If remission is not attained within three months, alternative treatments should be investigated.6
Conclusions
Because of the cost of medication and monitoring and the risk of serious drug side effects, treating dogs with PDH can be challenging. Therapy should only be instituted when clinical signs (i.e. Cushing's syndrome) are present. Generally, treatment of PDH is undertaken to improve the quality of life for the pet and the owner (e.g. improving polyuria, resolving dermatopathy) rather than to increase the length of the pet's life. We do not advocate medical therapy of PDH based on endocrinologic test results alone. Usually, therapy should be undertaken when clinical signs of Cushing's syndrome adversely affect quality of life. Treatment should also be instituted in circumstances in which PDH is exacerbating or causing other serious illnesses (e.g. diabetes mellitus, hypertension, thromboembolic disease, protein-losing nephropathy).
While many treatment options exist, mitotane remains the most effective, readily available therapy for PDH in the United States. The protocol for selective adrenocorticolysis with mitotane remains our typical choice for therapy. Trilostane is gaining acceptance as an alternative to mitotane, but it is not readily available. The cost of trilostane is generally higher than that of mitotane. In our hospital, trilostane is nearly twice the cost for an average-sized dog with PDH. Ketoconazole therapy can ameliorate signs of PDH, but the therapy may have greater utility in the short-term treatment of dogs with adrenal-dependent disease, pending adrenalectomy, than for long-term management of PDH. If diagnostic tests become available to allow differentiation of PDH due to pars intermedia tumors from the more common pars distalis tumors, L-deprenyl may be the safest treatment for this subset of patients. Regardless of the treatment chosen, frequent monitoring is critical to allow adjustment of therapy while minimizing adverse effects.
Amy E. DeClue, DVM
Leah A. Cohn, DVM, PhD, DACVIM
Marie E. Kerl, DVM, DACVIM, DACVECC
Department of Veterinary Medicine and Surgery
College of Veterinary Medicine
University of Missouri
Columbia, MO 65211
REFERENCES
1. Meij, B. et al.: Progress in transsphenoidal hypophysectomy for treatment of pituitary-dependent hyperadrenocorticism in dogs and cats. Mol. Cell Endocrinol. 197 (1-2):89-96; 1971.
2. Goossens, M.M. et al.: Efficacy of cobalt 60 radiotherapy in dogs with pituitary-dependent hyperadrenocorticism. JAVMA 212 (3):374-376; 1998.
3. Theon, A.P.; Feldman, E.C.: Megavoltage irradiation of pituitary macrotumors in dogs with neurologic signs. JAVMA 213 (2):225-231; 1998.
4. Feldman, E.; Nelson, R.W.: Canine hyperadrenocorticism (Cushing's Syndrome). Canine and Feline Endocrinology and Reproduction, 3rd Ed. W.B. Saunders, Philadelphia, Pa., 2004; pp 252-357.
5. Feldman, E.; Nelson, R.W.: Hypoadrenocorticism (Addison's Disease). Canine and Feline Endocrinology and Reproduction, 3rd Ed. W.B. Saunders, Philadelphia, Pa., 2004; pp 394-439.
6. Peterson, M.E.: Medical treatment of canine pituitary-dependent hyperadrenocorticism (Cushing's disease). Vet. Clin. North Am. (Small Anim. Pract.) 31 (5):1005-1015; 2001.
7. Peterson, M.E.: Medical treatment of pituitary-dependent hyperadrenocorticism in dogs: Should L-deprenyl (Anipryl) ever be used? J. Vet. Intern. Med. 13 (4):289-290; 1999.
8. Kintzer, P.P.; Peterson, M.E.: Mitotane (o,p'-DDD) treatment of 200 dogs with pituitary-dependent hyperadrenocorticism. J. Vet. Intern. Med. 5 (3):182-190; 1991.
9. Nelson, A.A. et al.: Severe adrenal cortical atrophy (cytotoxic) and hepatic damage produced in dogs by feeding 2,2-bis (para-chlorophenyl)-1,1-trichloroethane (DDD or TDE). Arch. Pathol. 48:387; 1949.
10. Schechter, R.D. et al.: Treatment of Cushing's syndrome in the dog with an adrenocorticolytic agent (o,p'DDD). JAVMA 162 (8):629-639; 1973.
11. Behrend, E.N. et al.: Treatment of hyperadrenocorticism in dogs: A survey of internists and dermatologists. JAVMA 215 (7):938-943; 1999.
12. Peterson, M.E.; Kintzer, P.P.: Medical treatment of pituitary-dependent hyperadrenocorticism. Mitotane. Vet. Clin. North Am. (Small Anim. Pract.) 27 (2):255-272; 1997.
13. Cai, W. et al.: Metabolic activation and binding of mitotane in adrenal cortex homogenates. J. Pharm. Sci. 84 (2):134-138; 1995.
14. Behrend, E. et al.: Medical therapy of canine Cushing's syndrome. Compend. Cont. Ed. 20 (2):679-696; 1998.
15. Arnold, D.L.: Mitotane. Compend. Cont. Ed. 23 (9):796-797, 815; 2001.
16. Plumb, D.C.: Mitotane, ketoconazole. Veterinary Drug Handbook (Pocket Edition), 4th Ed. Iowa State University, Ames, 2002; pp 588-590.
17. Kintzer, P.P.; Peterson, M.E.: Treatment and long-term follow-up of 205 dogs with hypoadrenocorticism. J. Vet. Intern. Med. 11 (2):43-49; 1997.
18. Nichols, R.: Complications and concurrent disease associated with canine hyperadrenocorticism. Vet. Clin. North Am. (Small Anim. Pract.) 27 (2):309-320; 1997.
19. Rijnberk, A.; Belshaw, B.E.: An alternative protocol for the medical management of canine pituitary-dependent hyperadrenocorticism. Vet. Rec. 122 (20):486-488; 1988.
20. den Hertog, E. et al.: Treatment of pituitary-dependent hyperadrenocorticism in the dog by non-selective adrenocorticolysis with o,p'-DDD. Vet. Q. 19 (suppl. 1):S17; 1997.
21. Rijnberk, A. et al.: O,p'-DDD treatment of canine hyperadrenocorticism: An alternative protocol. Kirk's Current Veterinary Therapy Small Animal Practice XI (R.W. Kirk; J.D. Bonagura, eds.). W.B. Saunders, Philadelphia, Pa., 1992; pp 345-349.
22. den Hertog, E. et al.: Results of non-selective adrenocorticolysis by o,p'-DDD in 129 dogs with pituitary-dependent hyperadrenocorticism. Vet. Rec. 144 (1):12-17; 1999.
23. Potts, G.O. et al.: Trilostane, an orally active inhibitor of steroid biosynthesis. Steroids 32 (2):257-267; 1978.
24. Neiger, R.: New treatments for Cushing's disease. Proc. Br. Small Anim. Vet. Assoc., BSAVA, Birmingham, U.K., 2003; pp 475-477.
25. Braddock, J.A. et al.: Trilostane treatment in dogs with pituitary-dependent hyperadrenocorticism. Aust. Vet. J. 81 (10):600-607; 2003.
26. Neiger, R. et al.: Trilostane treatment of 78 dogs with pituitary-dependent hyperadrenocorticism. Vet. Rec. 150 (26):799-804; 2002.
27. Ruckstuhl, N.S. et al.: Results of clinical examinations, laboratory tests, and ultrasonography in dogs with pituitary-dependent hyperadrenocorticism treated with trilostane. AJVR 63 (4):506-512; 2002.
28. Neiger, R. et al.: Trilostane therapy of canine hyperadrenocorticism. Proc. ACVIM, ACVIM, Dallas, Texas, 2002; pp 544-546.
29. Braddock, J.A.: Medical treatment of hyperadrenocorticism in the dog. Aust. Vet. J. 81 (1-2):31-33; 2003.
30. Bruyette, D. et al.: Ketoconazole and its use in the management of canine Cushing's disease. Compend. Cont. Ed. 10 (12):1379-1385; 1988.
31. Queiroz-Telles, F. et al.: Adrenal response to corticotrophin and testosterone during long-term therapy with itraconazole in patients with chromoblastomycosis. J. Antimicrob. Chemother. 40 (6):899-902; 1997.
32. Feldman, E.C. et al.: Plasma cortisol response to ketoconazole administration in dogs with hyperadrenocorticism. JAVMA 197 (1):71-78; 1990.
33. Kemppainen, R.J.; Sartin, J.L.: In vivo evidence for dopaminergic regulation of the canine pituitary intermediate lobe. Acta Endocrinol. 113 (4):471-478; 1986.
34. Peterson, M.E. et al.: Plasma immunoreactive proopiomelanocortin peptides and cortisol in normal dogs and dogs with Addison's disease and Cushing's syndrome: Basal concentrations. Endocrinology 119 (2):720-730; 1986.
35. Zerbe, C.A. et al.: Domperidone treatment enhances corticotropin-releasing hormone stimulated adrenocorticotropic hormones release from the dog pituitary. Neuroendocrinology 57 (2):282-288; 1993.
36. Bruyette, D.S. et al.: Treating canine pituitary-dependent hyperadrenocorticism with L-deprenyl. Vet. Med. 92 (8):711-727; 1997.
37. Cohn, L.A. et al.: Effects of selegiline, phenylpropanolamine, or a combination of both on physiologic and behavioral variables in healthy dogs. AJVR 63 (6):827-832; 2002.
38. Reusch, C.E. et al.: The efficacy of L-Deprenyl in dogs with pituitary-dependent hyperadrenocorticism. J. Vet. Intern. Med. 13 (4):291-301; 1999.
39. Milgram, N.W. et al.: Effects of chronic oral administration of L-deprenyl in the dog. Pharmacol. Biochem. Behav. 51 (2-3):421-428; 1995.
40. Bruyette, D.S. et al.: Management of canine pituitary-dependent hyperadrenocorticism with l-deprenyl (Anipryl). Vet. Clin. North Am. (Small Anim. Pract.) 27 (2):273-286; 1997.
Sweet pee new remedy in feline diabetes
November 9th 2023A novel class of drugs normalizes blood glucose in type 2 diabetic cats by dumping sugar into urine rather than modulating glucose uptake in the tissues but patient selection and close monitoring are crucial to using them safely
Read More